1,8-Octanediol
1,8-Octanediol[edit]
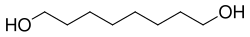
1,8-Octanediol is a diol with the chemical formula C_H__O_. It is a type of glycol that consists of an eight-carbon chain with hydroxyl groups (-OH) attached to the first and eighth carbon atoms. This compound is also known as octamethylene glycol.
Chemical Properties[edit]
1,8-Octanediol is a linear molecule with two hydroxyl groups, which makes it a bifunctional compound. The presence of these hydroxyl groups allows it to participate in various chemical reactions, such as esterification and etherification.
Physical Properties[edit]
1,8-Octanediol is a white crystalline solid at room temperature. It has a melting point of approximately 60°C and a boiling point of around 250°C. The compound is soluble in water and many organic solvents, which makes it useful in various industrial applications.
Applications[edit]
1,8-Octanediol is used in the production of polymers, plasticizers, and lubricants. It serves as a monomer in the synthesis of polyesters and polyurethanes. Additionally, it is used in the formulation of cosmetics and personal care products due to its moisturizing properties.
Synthesis[edit]
The synthesis of 1,8-Octanediol can be achieved through several methods, including the hydrogenation of suberic acid or its esters. Another method involves the reduction of 1,8-octanedione using suitable reducing agents.
Safety and Handling[edit]
1,8-Octanediol should be handled with care, as with all chemical substances. It is important to use appropriate personal protective equipment (PPE) when handling this compound to prevent skin and eye contact. In case of exposure, affected areas should be rinsed thoroughly with water.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian