Veratridine
A steroid-derived alkaloid toxin
| Chemical Compound | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
Veratridine is a steroid-derived alkaloid toxin that affects sodium channels in nerve cells. It is primarily found in the seeds of the Schoenocaulon plant, commonly known as sabadilla, and in the Liliaceae family of plants. Veratridine is known for its ability to increase the permeability of sodium channels, leading to prolonged depolarization of the nerve cell membrane.
Chemical structure[edit]
Veratridine is a complex molecule with a steroidal backbone. The structure of veratridine includes multiple hydroxyl groups and a distinctive arrangement of carbon rings. The molecular formula is C36H51NO11. The structure is depicted in the image
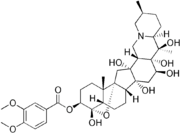
.
Mechanism of action[edit]
Veratridine acts by binding to the voltage-gated sodium channels on the neuronal cell membrane. This binding causes the channels to remain open longer than usual, which results in a continuous influx of sodium ions into the cell. The prolonged depolarization prevents the cell from returning to its resting state, leading to increased neuronal excitability. The specific binding site of veratridine on the sodium channel is illustrated in the image
.
Toxicity and effects[edit]
The toxic effects of veratridine are primarily due to its action on the nervous system. Symptoms of veratridine poisoning include nausea, vomiting, diarrhea, muscle weakness, and convulsions. In severe cases, it can lead to respiratory failure and death. The compound is used in research to study the function of sodium channels and the effects of their prolonged activation.
Applications in research[edit]
Veratridine is utilized in neuroscience research to investigate the properties of sodium channels and their role in neurotransmission. It serves as a tool to understand the mechanisms of nerve impulse propagation and the effects of channelopathies, which are disorders caused by dysfunctional ion channels.
Related compounds[edit]
Veratridine is structurally related to other steroidal alkaloids, such as veratrine and cevadine. These compounds share similar mechanisms of action and are also found in plants of the Liliaceae family. The comparison of steroid and veratridine ring backbones is shown in the image
.
Related pages[edit]
-
Veratridine structure
-
Steroid and Veratridine ring backbones
-
Veratridine binding site
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
