Triazolam
A benzodiazepine medication used for short-term treatment of insomnia
| Triazolam | |
|---|---|
|
|
Triazolam is a benzodiazepine medication primarily used for the short-term treatment of insomnia. It is known for its fast onset of action and short duration of effect. Triazolam is marketed under various brand names, with Halcion being one of the most well-known.
Medical uses[edit]
Triazolam is prescribed for the short-term management of insomnia, particularly in patients who have difficulty falling asleep. Due to its rapid onset, it is effective in reducing the time it takes to fall asleep. However, it is generally recommended for short-term use, typically not exceeding 7 to 10 days, due to the risk of dependence and other adverse effects.
Pharmacology[edit]
Triazolam works by enhancing the effect of the neurotransmitter gamma-aminobutyric acid (GABA) at the GABA_A receptor, which results in sedative, hypnotic, anxiolytic, anticonvulsant, and muscle relaxant properties. Its short half-life makes it suitable for use as a sleep aid, as it reduces the likelihood of next-day sedation.
Side effects[edit]
Common side effects of triazolam include drowsiness, dizziness, headache, and nausea. More serious side effects can include memory impairment, confusion, and hallucinations. Due to its potential for abuse and dependence, triazolam is classified as a Schedule IV controlled substance in the United States.
Contraindications[edit]
Triazolam is contraindicated in patients with a known hypersensitivity to benzodiazepines, as well as in those with myasthenia gravis, severe respiratory insufficiency, or sleep apnea. It should be used with caution in patients with a history of substance abuse or depression.
Interactions[edit]
Triazolam can interact with other medications that depress the central nervous system, such as alcohol, opioids, and other benzodiazepines, leading to increased sedation and respiratory depression. It is also metabolized by the cytochrome P450 enzyme system, particularly CYP3A4, and its effects can be altered by inhibitors or inducers of this enzyme.
History[edit]
Triazolam was first introduced in the 1970s and quickly became a popular treatment for insomnia due to its efficacy and short duration of action. However, concerns about its safety profile, particularly regarding its potential for abuse and adverse psychiatric effects, have led to more cautious prescribing practices.
Related pages[edit]
References[edit]
Triazolam[edit]
-
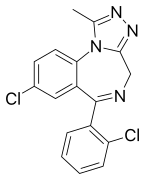
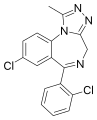
Chemical structure of Triazolam
-
Ball-and-stick model of Triazolam
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian