Strontium fluoride
A chemical compound of strontium and fluorine
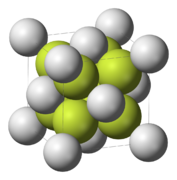
Strontium fluoride is an inorganic compound with the formula SrF_. It is a white crystalline solid that is slightly soluble in water. Strontium fluoride is used in a variety of applications, including optics and electronics.
Structure[edit]
Strontium fluoride crystallizes in the fluorite structure, which is a cubic arrangement. In this structure, each strontium ion is surrounded by eight fluoride ions, and each fluoride ion is surrounded by four strontium ions. This arrangement is similar to that of calcium fluoride, another compound with the fluorite structure.
Properties[edit]
Strontium fluoride is known for its high melting point and low solubility in water. It is a stable compound that does not react with most acids or bases. The compound is also transparent to a wide range of wavelengths, making it useful in optical applications.
Applications[edit]
Strontium fluoride is used in the production of optical materials, such as lenses and windows, due to its transparency and low refractive index. It is also used in the electronics industry as a component in certain types of semiconductors.
Production[edit]
Strontium fluoride can be produced by the reaction of strontium carbonate with hydrofluoric acid. The reaction produces strontium fluoride and carbon dioxide:
- SrCO_ + 2 HF _ SrF_ + CO_ + H_O
Safety[edit]
While strontium fluoride is generally considered to be of low toxicity, it should be handled with care. Inhalation or ingestion of the compound can be harmful, and appropriate safety measures should be taken when working with it.
Related pages[edit]
| Branches of chemistry | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian