Pyrogallol
Pyrogallol or pyrogallic acid is an organic compound with the formula C6H3(OH)3. It is a white, water-soluble solid although older samples can appear yellowish. Derived from gallic acid obtained from the gallnut, pyrogallol has found use in various applications ranging from photography to medicine. Its chemical structure consists of a benzene ring with three hydroxyl groups (−OH) positioned adjacently, making it a trihydroxybenzene.
History[edit]
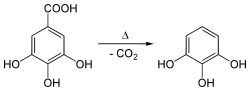
Pyrogallol was historically produced by heating gallic acid, a process that leads to the decarboxylation of gallic acid, releasing carbon dioxide (CO2) and forming pyrogallol. This method reflects the origin of its name, "pyro" referring to fire and "gallol" indicating its derivation from gallic acid. In the 19th century, pyrogallol played a crucial role in photographic development processes and was also used in hair dye formulations.
Chemical Properties[edit]
Pyrogallol is highly reactive towards oxygen, a property that has been exploited in various analytical chemistry applications, such as measuring oxygen content in gas mixtures. This reactivity is also the basis for its use as a reducing agent in photographic developer solutions. Pyrogallol's acidic nature allows it to form salts with metals, and its three hydroxyl groups can participate in complexation reactions with metal ions.
Applications[edit]
Photography[edit]
In photography, pyrogallol was one of the first developing agents used to develop photographic plates. Its strong reducing properties enable it to reduce silver ions to metallic silver, thereby forming the image. However, its use has declined with the advent of more stable and less toxic chemicals.
Analytical Chemistry[edit]
Pyrogallol has been utilized in analytical chemistry for the quantitative determination of oxygen. The method involves the absorption of oxygen by a solution of pyrogallol, changing the solution's properties in a measurable way.
Medicine[edit]
Historically, pyrogallol found applications in dermatology for treating skin conditions such as psoriasis and eczema. However, due to its potential toxicity and the development of safer alternatives, its use in medicine has significantly decreased.
Safety[edit]
Pyrogallol is toxic upon ingestion, inhalation, or skin contact, with the potential to cause severe health effects. It is also a strong reducing agent, which makes it a fire hazard when in contact with oxidizing substances. Safety measures include wearing protective equipment and ensuring proper ventilation when handling the compound.
Environmental Impact[edit]
The environmental impact of pyrogallol has not been extensively studied, but like many organic compounds, it is expected to biodegrade under certain conditions. However, its toxicity to aquatic life and potential to cause oxidative damage in the environment should not be overlooked.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
