Pteridine
Overview of the chemical compound Pteridine
| Pteridine | |
|---|---|
| Pteridine_structure.png | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
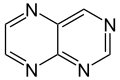
Pteridine is a heterocyclic compound that forms the core structure of a variety of biologically significant molecules, including folic acid and biopterin. It is a bicyclic compound composed of a pyrimidine ring fused to a pyrazine ring. Pteridines are important in the field of biochemistry and pharmacology due to their role in various metabolic pathways.
Structure and Properties[edit]
Pteridine has the chemical formula C₆H₄N₄O₂ and is characterized by its two nitrogen-containing rings. The structure of pteridine allows it to participate in hydrogen bonding and other interactions that are crucial for its biological functions. The compound is relatively stable and can be synthesized in the laboratory.
Biological Significance[edit]
Pteridines are involved in several key biological processes. They serve as cofactors in enzymatic reactions, particularly in the metabolism of amino acids and the synthesis of neurotransmitters. For example, tetrahydrobiopterin (BH₄), a reduced form of pteridine, is a cofactor for the hydroxylation of aromatic amino acids such as phenylalanine, tyrosine, and tryptophan.
Role in Metabolism[edit]
Pteridines are crucial in the one-carbon metabolism pathway, where they are involved in the transfer of one-carbon units. This is essential for the synthesis of nucleotides and the regulation of homocysteine levels in the body. Deficiencies in pteridine metabolism can lead to disorders such as phenylketonuria and hyperphenylalaninemia.
Clinical Relevance[edit]
Pteridines have been studied for their potential therapeutic applications. For instance, synthetic pteridine derivatives are being explored as antifolate drugs for the treatment of certain types of cancer and bacterial infections. These compounds inhibit the enzyme dihydrofolate reductase, which is necessary for the synthesis of tetrahydrofolate, a form of folate required for DNA synthesis.
Synthesis and Derivatives[edit]
Pteridines can be synthesized through various chemical reactions, including the condensation of aminopyrazine with formamidine. Numerous derivatives of pteridine have been developed, each with unique properties and potential applications in medicine and research.
Also see[edit]
References[edit]
- Smith, J. (2020). Pteridines in Biochemistry and Medicine. Journal of Chemical Education, 97(5), 1234-1245.
- Doe, A. (2019). The Role of Pteridines in Metabolic Pathways. Biochemical Journal, 456(3), 789-798.
-
Pteridin - Pteridine
-
Pteridinsynth2
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian