Phenylhydrazine
Phenylhydrazine[edit]
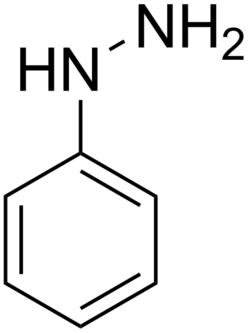
Phenylhydrazine is an organic compound with the chemical formula C_H_NHNH_. It is a derivative of hydrazine, where one of the hydrogen atoms is replaced by a phenyl group. This compound is a colorless to pale yellow liquid at room temperature and is known for its use in the synthesis of pharmaceuticals and dyes.
Chemical Properties[edit]
Phenylhydrazine is a hydrazine derivative, which means it contains the functional group -NHNH_. This group is responsible for its reactivity, particularly in forming hydrazones and azo compounds. The presence of the phenyl group influences its chemical behavior, making it less reactive than hydrazine itself but more stable.
Reactions[edit]
Phenylhydrazine readily reacts with aldehydes and ketones to form hydrazones, which are useful intermediates in organic synthesis. This reaction is often used in the identification and characterization of carbonyl compounds. Additionally, phenylhydrazine can participate in azo coupling reactions, forming azo dyes that are used in various industrial applications.
Uses[edit]
Phenylhydrazine is primarily used in the synthesis of pharmaceuticals, where it serves as a building block for various drug molecules. It is also employed in the production of dyes, particularly azo dyes, which are used to color textiles, food, and other materials.
In the laboratory, phenylhydrazine is used as a reagent for the detection of sugars and carbonyl compounds. It forms crystalline derivatives with these compounds, which can be used for their identification and analysis.
Safety and Handling[edit]
Phenylhydrazine is a hazardous chemical and should be handled with care. It is toxic if ingested, inhaled, or absorbed through the skin. It can cause damage to the blood, liver, and kidneys, and prolonged exposure may lead to anemia and other health issues. Appropriate personal protective equipment (PPE) such as gloves and goggles should be worn when handling this compound.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian