Oxygen compounds


Oxygen compounds are chemical substances that contain oxygen atoms bonded to other elements. Oxygen is highly reactive and forms a variety of compounds with metals and nonmetals. It is essential for life on Earth and participates in numerous chemical reactions, including combustion, oxidation, and biological processes like cellular respiration.
Types of Oxygen Compounds[edit]
Oxygen compounds can be broadly categorized based on the types of elements they bond with and their chemical structures. These include:
Oxides[edit]
Oxides are compounds formed by the reaction of oxygen with another element. They can be classified as:
- Basic oxides: Formed with metals, such as calcium oxide (CaO) and magnesium oxide (MgO).
- Acidic oxides: Formed with nonmetals, such as carbon dioxide (CO₂) and sulfur trioxide (SO₃).
- Amphoteric oxides: Exhibit both acidic and basic properties, such as aluminum oxide (Al₂O₃).
- Neutral oxides: Neither acidic nor basic, such as nitrous oxide (N₂O).
Peroxides[edit]
Peroxides contain an oxygen-oxygen single bond (O-O), which is highly reactive. Examples include:
- Hydrogen peroxide (H₂O₂), a common disinfectant and bleaching agent.
- Sodium peroxide (Na₂O₂), used in industrial and chemical applications.
Superoxides[edit]
Superoxides contain the superoxide anion (O₂⁻). These compounds are often unstable and highly reactive. Examples include:
- Potassium superoxide (KO₂), used in oxygen masks and chemical oxygen generators.
Hydroxides[edit]
Hydroxides contain the hydroxyl group (OH⁻) and are commonly found in bases. Examples include:
- Sodium hydroxide (NaOH), also known as lye, widely used in industry.
- Calcium hydroxide (Ca(OH)₂), used in construction and as a neutralizing agent.
Ozonides[edit]
Ozonides are compounds containing the ozone anion (O₃⁻). These are less common and often highly unstable.
Organic Oxygen Compounds[edit]
Oxygen is a critical element in many organic compounds:
- Alcohols: Contain the hydroxyl (-OH) group, such as ethanol (C₂H₅OH).
- Ethers: Contain an oxygen atom bonded to two alkyl or aryl groups, such as dimethyl ether (CH₃OCH₃).
- Carbonyl compounds: Include aldehydes and ketones, such as formaldehyde (HCHO) and acetone (CH₃COCH₃).
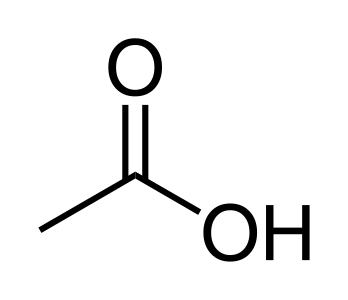
- Carboxylic acids: Contain the carboxyl group (-COOH), such as acetic acid (CH₃COOH).
- Esters andanhydrides: Derived from carboxylic acids, such as methyl acetate.
Reactions Involving Oxygen Compounds[edit]
Oxygen compounds participate in numerous chemical reactions, including:
- Combustion: The reaction of oxygen with a fuel to produce energy, as seen in the burning of hydrocarbons.
- Oxidation: The loss of electrons from a substance, often involving oxygen as the oxidizing agent.
- Reduction: The gain of electrons, where oxygen compounds like oxides may be reduced to their elemental form.
- Acid-base reactions: Many oxygen compounds, such as oxides and hydroxides, play a role in neutralization reactions.
Applications of Oxygen Compounds[edit]
Oxygen compounds have a wide range of applications in industry, medicine, and daily life:
- Industrial uses: Hydrogen peroxide is used as a bleaching agent, and calcium oxide is critical in cement production.
- Medical uses: Ozone therapy and oxygenated water are explored for therapeutic benefits.
- Environmental applications: Oxygen compounds like ozone (O₃) protect the Earth from ultraviolet radiation in the ozone layer.
Biological Importance[edit]
Oxygen compounds are essential to life:
- Water (H₂O) is the most vital compound for all living organisms.
- Oxygen is a key reactant in aerobic respiration.
- Organic oxygen compounds, such as carbohydrates and lipids, are crucial energy sources for living organisms.
Safety and Hazards[edit]
Many oxygen compounds are reactive and require careful handling:
- Flammability: Peroxides and superoxides can be highly explosive.
- Corrosivity: Compounds like sodium hydroxide and sulfuric acid can cause severe burns.
- Toxicity: Excessive exposure to compounds like carbon monoxide (CO) can be fatal.
Gallery[edit]
-
Water, the most essential oxygen compound for life.
-
Carbon dioxide (CO₂), a critical greenhouse gas.
-
Calcium oxide.
-
Potassium superoxide powder.
-
Acetic-acid (CH₃COOH), found in vinegar.
-
Quartz, Tibet
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian