Redox
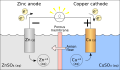
Redox (short for reduction–oxidation reaction) is a type of chemical reaction in which the oxidation states of atoms are changed. Redox reactions are characterized by the transfer of electrons between chemical species, most often with one species (the reductant) undergoing oxidation (losing electrons) while another species (the oxidant) undergoes reduction (gains electrons).
Overview[edit]
The term 'redox' comes from two concepts involved with electron transfer: 'reduction' and 'oxidation'. It can be explained in simple terms:
- Oxidation is the loss of electrons or an increase in the oxidation state of an atom, an ion, or of certain atoms in a molecule.
- Reduction is the gain of electrons or a decrease in the oxidation state of an atom, an ion, or of certain atoms in a molecule.
Redox reactions[edit]
In redox processes, the reductant transfers electrons to the oxidant. Thus, in the reaction, the reductant or reducing agent loses electrons and is oxidized, and the oxidant or oxidizing agent gains electrons and is reduced.
Examples[edit]
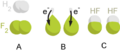
The classic redox reaction is a reaction between hydrogen and fluorine: H2 + F2 → 2HF In this reaction, hydrogen is oxidized because it loses its electron to fluorine, which is reduced.
See also[edit]
References[edit]
-
NaF
-
Reaction between strong oxidizing and reducing agent
-
GHS pictogram for flammable
-
Redox reaction
-
Galvanic cell with no cation flow
-
Rust screw
-
PyOx
-
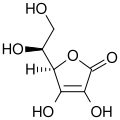
Ascorbic acid structure
-
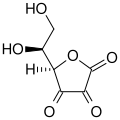
Dehydroascorbic acid
-
Extremely overripe banana
-
Vysoke Pece
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian