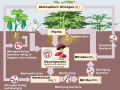
Nitrogen fixation
Nitrogen fixation is a process by which nitrogen in the Earth's atmosphere is converted into ammonia (NH3) or related nitrogenous compounds. Atmospheric nitrogen, is molecular dinitrogen (N2), a relatively nonreactive molecule that is metabolically useless to all but a few microorganisms. Biological nitrogen fixation converts N2 into ammonia, which is metabolized by most organisms. Nitrogen fixation is essential for life because fixed inorganic nitrogen compounds are required for the biosynthesis of all nitrogen-containing organic compounds, such as amino acids and proteins, nucleoside triphosphates and nucleic acids. As part of the nitrogen cycle, it is essential for agriculture and the manufacture of fertilizer. It is also, indirectly, relevant to the manufacture of all chemical compounds that contain nitrogen, which includes some explosives, pharmaceuticals, and dyes.
Process[edit]
Nitrogen fixation is carried out naturally in the soil by microorganisms termed diazotrophs that include bacteria such as Azotobacter and archaea. Some nitrogen-fixing bacteria have symbiotic relationships with plant groups, especially legumes. Looser non-symbiotic relationships between diazotrophs and plants are often important in soil fertility.
Industrial nitrogen fixation[edit]
Humans also practice nitrogen fixation as a means to fertilize soils. Industrial nitrogen fixation is achieved through the Haber-Bosch process, which utilizes high temperatures and pressures to convert nitrogen gas and hydrogen into ammonia.
Environmental impact[edit]
Nitrogen fixation, both natural and human-induced, has environmental impacts. Nitrogen-rich compounds derived from nitrogen fixation can cause a form of pollution known as eutrophication, a harmful overgrowth of plant life.
See also[edit]
Nitrogen_fixation[edit]
-
Diagram of the nitrogen cycle
-
Root nodules on fava bean plant
-
Sectioned alder root nodule gall
-
D. C. Bardwell Study of Nitrogen Fixation
-
Lightning as a natural source of nitrogen fixation
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian