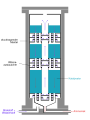
Haber process
Haber Process is a method of synthesizing ammonia from hydrogen and nitrogen. Developed by Fritz Haber in the early 20th century, the process is important in the large-scale production of fertilizers and explosives.
History[edit]
The Haber process was developed in the early 20th century by German chemist Fritz Haber. Haber was awarded the Nobel Prize in Chemistry in 1918 for this development. The process was later commercialized by Carl Bosch, leading to its widespread use in the industrial production of ammonia.
Process[edit]
The Haber process involves the direct synthesis of ammonia from its elements, hydrogen and nitrogen. The reaction is carried out at high temperatures and pressures, and is typically facilitated by an iron catalyst.
The overall reaction is: N2 + 3H2 ⇌ 2NH3
This reaction is exothermic, releasing heat and thus favoring the formation of ammonia at lower temperatures. However, the reaction rate is slow at lower temperatures, so the process is typically carried out at a high temperature to increase the reaction rate.
Industrial significance[edit]
The Haber process is of great industrial significance as it allows for the large-scale production of ammonia. Ammonia is a key ingredient in the production of fertilizers, which are essential for modern agriculture. It is also used in the production of nitric acid, which is used in the manufacture of explosives.
Environmental impact[edit]
While the Haber process has greatly increased the availability of fertilizers and thus contributed to increased agricultural productivity, it also has significant environmental impacts. The process requires large amounts of energy, typically derived from fossil fuels, contributing to greenhouse gas emissions. Additionally, the widespread use of nitrogen-based fertilizers has led to increased nitrogen levels in water bodies, contributing to eutrophication and the degradation of aquatic ecosystems.
See also[edit]
Haber process[edit]
-
Fritz Haber
-
Carl Bosch (1908)
-
Ammonia Reactor BASF
-
Steam Methane Reforming and Water Gas Shift
-
Ammonia Reactor 1913 Oppau
-
Heterogeneous Catalysis
-
Haber-Bosch Process Diagram
-
Ammonia Reactor Diagram
-
Catalyst for Ammonia Synthesis
-
Potential Energy Diagram for Ammonia Synthesis
-
Severnside Fertilizer Works
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian