Lapyrium
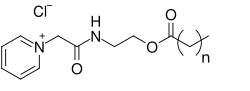
== Lapyrium ==
Lapyrium is a chemical compound that belongs to the class of quaternary ammonium compounds. It is primarily used as an antiseptic and disinfectant in various medical and industrial applications. Lapyrium is known for its effectiveness in killing a wide range of microorganisms, including bacteria, viruses, and fungi.
Chemical Properties[edit]
Lapyrium has a molecular formula of C12H28ClN and a molecular weight of 221.82 g/mol. It is a colorless to pale yellow liquid with a characteristic odor. The compound is soluble in water and exhibits strong surface-active properties, making it an effective surfactant.
Uses[edit]
Lapyrium is widely used in the following applications:
- **Medical Field**: As an antiseptic for wound care and surgical instrument disinfection.
- **Industrial Applications**: In the formulation of cleaning agents and sanitizers for various surfaces.
- **Agriculture**: As a disinfectant in animal husbandry to prevent the spread of infectious diseases.
Mechanism of Action[edit]
Lapyrium works by disrupting the cell membranes of microorganisms, leading to cell lysis and death. This mechanism is similar to other quaternary ammonium compounds, which are known for their ability to denature proteins and inactivate enzymes essential for microbial survival.
Safety and Handling[edit]
While Lapyrium is effective as a disinfectant, it should be handled with care. Prolonged exposure can cause skin irritation and respiratory issues. It is recommended to use protective gear, such as gloves and masks, when handling the compound.
Related Compounds[edit]
Lapyrium is related to other quaternary ammonium compounds such as benzalkonium chloride, cetylpyridinium chloride, and didecyldimethylammonium chloride. These compounds share similar properties and uses but may vary in their spectrum of activity and toxicity.
See Also[edit]
- Antiseptic
- Disinfectant
- Quaternary ammonium compound
- Benzalkonium chloride
- Cetylpyridinium chloride
- Didecyldimethylammonium chloride
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian