Didecyldimethylammonium chloride
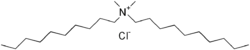
Didecyldimethylammonium chloride (DDAC) is a type of quaternary ammonium compound that is widely used as an antiseptic, disinfectant, and surfactant. It is a colorless liquid that is soluble in water, alcohol, and acetone.
Chemical Structure[edit]
DDAC is a cationic surfactant with the chemical formula C22H48ClN. It consists of two decyl chains (C10H21) attached to a nitrogen atom, which is also bonded to two methyl groups (CH3) and a chloride ion (Cl-). The presence of the positively charged nitrogen atom allows DDAC to interact with negatively charged surfaces, which is key to its antiseptic and disinfectant properties.
Uses[edit]
DDAC is primarily used as an active ingredient in a variety of disinfectant and antiseptic products. It is effective against a broad spectrum of microorganisms, including bacteria, fungi, and viruses. In addition to its antimicrobial properties, DDAC also has surfactant properties, which means it can reduce surface tension and enhance the cleaning ability of a product.
In the healthcare industry, DDAC is used in hard surface disinfectants, hand sanitizers, and wound antiseptics. In the food industry, it is used in sanitizing solutions for food contact surfaces. It is also used in water treatment, wood preservation, and in the textile industry for its softening and antistatic effects.
Safety[edit]
DDAC is generally considered safe for use in the concentrations found in consumer products. However, it can cause skin and eye irritation, and prolonged or repeated exposure can lead to respiratory irritation. It is recommended to use products containing DDAC in a well-ventilated area and to avoid direct contact with the skin and eyes.
Environmental Impact[edit]
DDAC is not readily biodegradable and can accumulate in the environment, particularly in aquatic ecosystems. It is toxic to aquatic life and can cause long-term adverse effects. Therefore, it is important to manage its use and disposal properly to minimize its environmental impact.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian