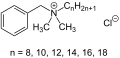
Quaternary ammonium cation
Quaternary ammonium cations, often abbreviated as Quats, are a class of ammonium compounds in which four organic groups are covalently bonded to a nitrogen atom, resulting in a positively charged ion or cation. These compounds are characterized by the general formula R4N+, where R represents an alkyl or aryl group. Due to their cationic nature, quaternary ammonium compounds exhibit unique properties, making them valuable in various industrial, medical, and environmental applications.
Properties[edit]
Quaternary ammonium cations are known for their surfactant properties, which make them effective as disinfectants and antiseptics. They disrupt microbial cell membranes, leading to cell death, and are effective against a wide range of bacteria, viruses, fungi, and algae. Their efficacy, however, can be influenced by factors such as temperature, pH, and the presence of organic matter.
Applications[edit]
Disinfectants and Antiseptics[edit]
Quats are widely used in healthcare settings, including hospitals and dental offices, for the disinfection of surfaces and instruments. Products containing quaternary ammonium compounds, such as benzalkonium chloride and cetrimonium bromide, are also used in personal care items like shampoos and conditioners, due to their antimicrobial properties and ability to condition hair.
Water Treatment[edit]
In water treatment processes, quaternary ammonium cations serve as algicides and biocides, controlling the growth of algae and other microorganisms in water systems, including cooling towers and swimming pools.
Agriculture[edit]
Quats are used in agriculture as pesticide components, helping to protect crops from fungal and bacterial diseases. They are also employed in animal husbandry for disinfecting livestock enclosures and equipment.
Safety and Environmental Concerns[edit]
While quaternary ammonium compounds are generally considered safe for human use, prolonged exposure or high concentrations can cause skin and respiratory irritation. Environmental concerns have been raised regarding their persistence and toxicity in aquatic environments, as they can be harmful to aquatic life.
Chemical Structure and Reactivity[edit]
The structure of quaternary ammonium cations allows for a wide range of chemical reactivity, depending on the nature of the R groups attached to the nitrogen atom. This versatility enables the synthesis of compounds tailored for specific applications, from phase transfer catalysts in organic synthesis to active ingredients in fabric softeners.
See Also[edit]
Quaternary_ammonium_cation[edit]
-
Quaternary ammonium cation
-
OBINIXBu4N1
-
OBINIXBu4N2
-
Benzalkonium chloride Structure V.1
-
Ion exchange resin beads
-
C18x2Me2Cl
-
Redrawn diesterquat salt (methanesulfonate anion) related to fabric softeners
-
AltDiesterquatCl
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian