Iodate
An article about the iodate ion in chemistry
Iodate[edit]
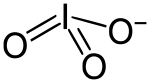
The iodate ion is a polyatomic ion with the chemical formula IO__. It is the conjugate base of iodic acid (HIO_) and is commonly encountered in various chemical reactions and compounds. The iodate ion is an important species in inorganic chemistry and is known for its oxidizing properties.
Structure[edit]
The iodate ion consists of one iodine atom centrally bonded to three oxygen atoms in a trigonal pyramidal arrangement. The iodine atom is in the +5 oxidation state, and the ion carries a net charge of -1. The geometry of the iodate ion is similar to that of the chlorate and bromate ions, which are also oxyanions of the halogens.
Properties[edit]
Iodate ions are strong oxidizing agents and can participate in various redox reactions. They are stable in aqueous solutions and can be reduced to iodide ions (I_) or oxidized to periodate ions (IO__) under appropriate conditions. The iodate ion is less reactive than the hypoiodite (IO_) and iodite (IO__) ions but more reactive than the periodate ion.
Occurrence[edit]
Iodate ions are found naturally in some minerals and are also produced industrially. They are used in the production of iodized salt, where potassium iodate (KIO_) is added to table salt to prevent iodine deficiency.
Uses[edit]
Iodate compounds are used in various applications, including:
- As an oxidizing agent in analytical chemistry.
- In the food industry as a source of iodine in iodized salt.
- In the synthesis of other iodine-containing compounds.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian