Chlorate
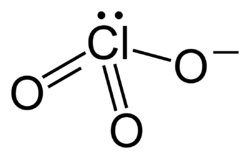
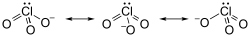
Chlorates are chemical compounds containing the chlorate ion, ClO3−. They are formed by the oxidation of chlorides in the presence of a strong oxidizing agent. Chlorates are powerful oxidizers and have been used in a variety of applications, from pyrotechnics to herbicides and as a source of oxygen in chemical oxygen generators.
Properties and Structure[edit]
The chlorate ion is a polyatomic ion with the chemical formula ClO3−. It consists of one chlorine atom covalently bonded to three oxygen atoms. The chlorine atom is in the +5 oxidation state. Chlorates are generally soluble in water, and their solubility increases with temperature.
Production[edit]
Chlorates can be produced through several methods. The most common method is the electrolysis of sodium chloride (salt) solution, which produces sodium chlorate, NaClO3. This process, known as the Hooker process, involves the oxidation of chloride ions to chlorate ions at the anode in an electrolytic cell.
Applications[edit]
Pyrotechnics[edit]
Chlorates are widely used in the pyrotechnics industry due to their strong oxidizing properties. They are used to produce fireworks, flares, and matches. However, their use has been reduced in favor of perchlorates, which are more stable and less sensitive to friction and impact.
Agriculture[edit]
In agriculture, sodium chlorate has been used as a non-selective herbicide. It is effective in killing a wide range of plants by disrupting the photosynthesis process. However, its use has declined due to environmental concerns and the availability of more selective herbicides.
Chemical Oxygen Generators[edit]
Chlorates are used in chemical oxygen generators, which are devices that release oxygen via a chemical reaction. These generators are used in submarines, aircraft, and space stations to provide an emergency supply of oxygen.
Safety and Environmental Concerns[edit]
Chlorates are strong oxidizers and can pose significant safety risks. They can react violently with combustible materials and are sensitive to friction and impact, which can lead to explosions. Proper handling and storage are essential to prevent accidents.
Environmental concerns are also associated with the use of chlorates. As herbicides, they can contaminate water sources and harm non-target plant species. The production and disposal of chlorates must be managed carefully to minimize environmental impact.
Regulation[edit]
Due to their potential health and environmental risks, the use and disposal of chlorates are regulated in many countries. Regulations may include restrictions on their use in certain applications, requirements for safe handling and storage, and guidelines for disposal.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
