Dimethyl trisulfide
Chemical compound
| Dimethyl trisulfide | |
|---|---|
| Dimethyl trisulfide.png | |
| Identifiers | |
| CAS Number | |
| PubChem CID | 12230 |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | C_2H_6S_3 |
| Molar Mass | |
| Appearance | Colorless to pale yellow liquid |
| Density | 1.157 g/cm³ |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
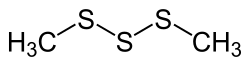
Dimethyl trisulfide (DMTS) is an organosulfur compound with the formula (CH3S)2S. It is a colorless to pale yellow liquid with a strong odor. DMTS is a significant component of the aroma of cooked cabbage and is also found in cruciferous vegetables such as broccoli and Brussels sprouts. It is also present in beer, cheese, and some wines.
Chemical Properties[edit]
Dimethyl trisulfide is part of a group of compounds known as polysulfides. It is characterized by the presence of three sulfur atoms in a chain, flanked by two methyl groups. The chemical structure can be represented as CH3-S-S-S-CH3. It has a molecular weight of 126.26 g/mol and a density of 1.157 g/cm³. DMTS has a melting point of -85°C and a boiling point of 151°C. It is slightly soluble in water but more soluble in organic solvents.
Occurrence[edit]
DMTS is naturally occurring and can be found in various foods and beverages. It is a key contributor to the flavor profile of cooked cabbage and other cruciferous vegetables. It is also found in beer, cheese, and some wines. The compound is produced during the thermal degradation of glucosinolates, which are sulfur-containing compounds found in these vegetables.
Applications[edit]
Dimethyl trisulfide is used in the food industry as a flavoring agent due to its strong odor and flavor characteristics. It is also used in the production of pesticides and as a chemical intermediate in the synthesis of other sulfur-containing compounds.
Safety and Handling[edit]
DMTS should be handled with care due to its strong odor and potential toxicity. It is advisable to use appropriate personal protective equipment (PPE) when handling this compound. In case of contact with skin or eyes, it is recommended to rinse thoroughly with water and seek medical attention if necessary.
Related Compounds[edit]
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
