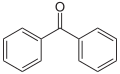
Benzophenone
Benzophenone is an organic compound with the formula (C6H5)2CO, often abbreviated as Ph2CO. It is a widely used building block in organic chemistry, being the parent diarylketone.
Structure and properties[edit]
Benzophenone is a white, crystalline solid with a sweet, rose-like aroma. It is poorly soluble in water but is well soluble in common organic solvents. It features a carbonyl group (C=O) bonded to two phenyl groups. The molecular geometry around the carbonyl is trigonal planar, but the molecule is not planar.
Production[edit]
Benzophenone can be produced by several methods. The most common method is the Friedel-Crafts acylation of benzene with benzoyl chloride in the presence of a Lewis acid such as aluminium chloride.
Uses[edit]
Benzophenone is a common photosensitizer in photochemistry. It crosses from the S1 state into the triplet state with nearly 100% yield. The resulting diradical will abstract a hydrogen atom from a suitable hydrogen donor to form a ketyl radical.
Benzophenone is used in manufacturing perfumes and soaps, to enhance their scent. It is also used in the rubber industry to prevent ultraviolet light from damaging rubber products.
Health effects[edit]
Benzophenone can cause skin and eye irritation. Long-term exposure may lead to more serious health problems such as liver and kidney damage. It is classified as a possible human carcinogen by the International Agency for Research on Cancer.
See also[edit]
References[edit]
Benzophenone[edit]
-
Benzophenon.svg
-
Benzophenone from xtal stable phase 1968 3D balls
-
Benzophenone from xtal stable phase 1968 3D vdW
-
Benzophenone vial
-
Haller-Bauer reaction
-
Solvent pot
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian