Benzoyl chloride

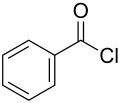
Benzoyl chloride, also known as benzenecarbonyl chloride, is an organic compound with the formula C6H5COCl. It is a colorless, sharp-smelling liquid, which fumes in air and has a strongly irritating effect on the skin and mucous membranes. Benzoyl chloride is a member of the acyl chlorides, a subclass of acid halides, and is widely used in organic synthesis.
Properties[edit]
Benzoyl chloride is characterized by its strong, pungent odor and its corrosive nature. It is less dense than water, with a density of 1.212 g/cm3, and it boils at 197.2 °C. Being an acyl chloride, it reacts vigorously with water, releasing hydrochloric acid (HCl) and forming benzoic acid.
Production[edit]
The primary method for producing benzoyl chloride involves the chlorination of benzoic acid in the presence of agents such as thionyl chloride, phosphorus trichloride, or phosphorus pentachloride. The reaction with thionyl chloride can be represented as follows:
C6H5COOH + SOCl2 → C6H5COCl + SO2 + HCl
This method is preferred for its high yields and the ease of removing the by-products, sulfur dioxide and hydrochloric acid, by simple evaporation.
Uses[edit]
Benzoyl chloride is primarily used in the synthesis of other organic compounds. It is a key starting material in the production of benzoyl peroxide, a common ingredient in acne treatments. It also finds application in the synthesis of dyes, fragrances, and pharmaceuticals, where it is used to introduce the benzoyl group into various molecules.
In addition to its use in chemical synthesis, benzoyl chloride can serve as a reagent in the preparation of esters and amides of benzoic acid, through reaction with alcohols and amines, respectively. These reactions typically require the use of a base to neutralize the hydrochloric acid formed as a byproduct.
Safety[edit]
Benzoyl chloride is a highly corrosive substance that can cause severe burns upon contact with skin and can be damaging to the eyes, respiratory system, and mucous membranes. Appropriate safety measures, including the use of personal protective equipment (PPE) such as gloves, goggles, and protective clothing, are essential when handling this chemical. It should be used only in well-ventilated areas or under a fume hood.
Environmental Impact[edit]
Due to its reactivity and the production of hydrochloric acid upon exposure to water, benzoyl chloride poses a risk to aquatic life and the environment. Measures should be taken to prevent its release into the environment and to ensure safe disposal in accordance with local regulations.
-
Benzoyl chloride 200
-
Benzoyl chloride from crystal 3D bs 17
-
Benzoyl chloride from crystal 3D sf
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian