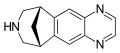
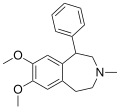
Benzazepine
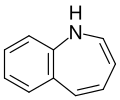
Benzazepine is a standard parent hydride of organic compounds that contain a benzene ring fused to an azepine. The fusion can occur in three different ways, leading to three parent structures. Benzazepines are a class of compounds in medicinal chemistry with a seven-membered heterocyclic ring containing one nitrogen atom, fused to a benzene ring.
Structure and Bonding[edit]
Benzazepines are characterized by a seven-membered ring structure with a nitrogen atom. The nitrogen atom is typically sp3 hybridized, meaning it forms three sigma bonds and one pi bond. The benzene ring is a six-membered ring with alternating single and double bonds, which gives it a high degree of aromaticity.
Synthesis[edit]
Benzazepines can be synthesized through several methods. One common method is the intramolecular Heck reaction, which involves the palladium-catalyzed coupling of an aryl halide and an alkene. Other methods include the Bischler-Napieralski reaction and the Pictet-Spengler reaction.
Applications[edit]
Benzazepines have a wide range of applications in medicinal chemistry. They are used as the core structure in many pharmaceutical drugs, including antipsychotics, antidepressants, and antihypertensives. Some well-known drugs that contain a benzazepine ring include Clozapine, Lorazepam, and Diltiazem.
See Also[edit]
- Azepine
- Benzene
- Medicinal Chemistry
- Heck Reaction
- Bischler-Napieralski reaction
- Pictet-Spengler reaction
References[edit]
-
Benzazepine
-
Benzazepine 3D balls
-
Benazepril structure
-
Fenoldopam
-
GSK-189,254
-
Ivabradine
-
Semagacestat structure
-
Varenicline
-
Trepipam
-
SKF38393
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian