Beckmann rearrangement
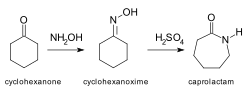




Beckmann rearrangement
The Beckmann rearrangement is an important organic reaction used in chemistry to convert an oxime to an amide under acidic conditions. This reaction is named after the German chemist Ernst Otto Beckmann, who first described it in 1886.
Mechanism[edit]
The Beckmann rearrangement involves the conversion of an oxime, which is typically derived from a ketone or aldehyde, into an amide. The reaction proceeds through the following steps:
1. Protonation of the oxime: The oxime is protonated by an acid, making the nitrogen more electrophilic. 2. Migration of the alkyl group: One of the alkyl groups adjacent to the oxime migrates to the nitrogen, forming a nitrilium ion. 3. Hydrolysis: The nitrilium ion is hydrolyzed to form the corresponding amide.
Applications[edit]
The Beckmann rearrangement is widely used in the synthesis of various pharmaceuticals, polymers, and other organic compounds. One of the most notable applications is the industrial production of caprolactam, which is a precursor to Nylon 6.
Reagents and Conditions[edit]
The reaction typically requires a strong acid such as sulfuric acid, hydrochloric acid, or phosphoric acid. In some cases, Lewis acids like aluminum chloride or phosphorus pentachloride can also be used to facilitate the rearrangement.
Examples[edit]
1. Cyclohexanone oxime to caprolactam: This is a classic example where cyclohexanone oxime undergoes Beckmann rearrangement to form caprolactam, which is then polymerized to produce Nylon 6. 2. Acetophenone oxime to acetanilide: Another example is the conversion of acetophenone oxime to acetanilide.
Related Reactions[edit]
The Beckmann rearrangement is related to several other organic reactions, including:
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian