Friedel–Crafts reaction
Friedel–Crafts Reaction[edit]
The Friedel–Crafts reaction is a set of reactions developed by Charles Friedel and James Crafts in 1877 to attach substituents to an aromatic ring. These reactions are of two main types: Friedel–Crafts alkylation and Friedel–Crafts acylation.
Friedel–Crafts Alkylation[edit]
The Friedel–Crafts alkylation involves the alkylation of an aromatic ring with an alkyl halide using a strong Lewis acid catalyst such as aluminum chloride (AlCl₃). This reaction is used to introduce an alkyl group into an aromatic compound.

Mechanism[edit]
The mechanism of the Friedel–Crafts alkylation involves the formation of a carbocation intermediate. The Lewis acid catalyst helps to generate the carbocation from the alkyl halide, which then attacks the aromatic ring to form the alkylated product.
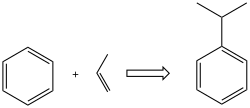
Limitations[edit]
Friedel–Crafts alkylation has several limitations, including carbocation rearrangement, polyalkylation, and the deactivation of the aromatic ring by electron-withdrawing groups.
Friedel–Crafts Acylation[edit]
The Friedel–Crafts acylation involves the acylation of an aromatic ring with an acyl chloride or acid anhydride in the presence of a Lewis acid catalyst. This reaction introduces an acyl group into the aromatic compound.
Mechanism[edit]
The mechanism of Friedel–Crafts acylation involves the formation of an acylium ion, which is a more stable electrophile than a carbocation. The acylium ion attacks the aromatic ring to form the acylated product.
Advantages[edit]
Friedel–Crafts acylation does not suffer from polyacylation because the acyl group is electron-withdrawing, which deactivates the aromatic ring towards further electrophilic substitution.
Applications[edit]
Friedel–Crafts reactions are widely used in the synthesis of aromatic ketones, alkylbenzenes, and other aromatic compounds. They are important in the production of pharmaceuticals, fragrances, and polymers.
Related Reactions[edit]
Friedel–Crafts reactions are related to other electrophilic aromatic substitution reactions such as nitration, sulfonation, and halogenation.

Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian