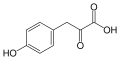
4-Hydroxyphenylpyruvic acid
An intermediate in the metabolism of the amino acid phenylalanine
| Chemical Compound | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
4-Hydroxyphenylpyruvic acid is an intermediate in the metabolism of the amino acid phenylalanine. It plays a crucial role in the tyrosine degradation pathway and is involved in several metabolic processes.
Structure and Properties[edit]
4-Hydroxyphenylpyruvic acid is a keto acid with the chemical formula C9H8O4. It contains a hydroxy group attached to the phenyl ring, which is characteristic of its structure. The presence of both a carboxylic acid group and a keto group makes it a key intermediate in various biochemical reactions.
Biosynthesis[edit]
4-Hydroxyphenylpyruvic acid is synthesized from phenylalanine through a series of enzymatic reactions. The first step involves the conversion of phenylalanine to tyrosine by the enzyme phenylalanine hydroxylase. Tyrosine is then converted to 4-hydroxyphenylpyruvic acid by the enzyme tyrosine aminotransferase.
Metabolic Pathway[edit]
In the tyrosine catabolism pathway, 4-hydroxyphenylpyruvic acid is further metabolized to homogentisic acid by the enzyme 4-hydroxyphenylpyruvate dioxygenase. This step is crucial for the breakdown of tyrosine and the production of energy and other metabolites.
Clinical Significance[edit]
Deficiencies or malfunctions in the enzymes involved in the metabolism of 4-hydroxyphenylpyruvic acid can lead to metabolic disorders. For example, a deficiency in 4-hydroxyphenylpyruvate dioxygenase can result in tyrosinemia type III, a rare genetic disorder characterized by elevated levels of tyrosine and its metabolites in the blood.
Related Compounds[edit]
Related Pages[edit]
-
4-Hydroxyphenylpyruvic acid
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian