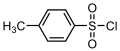
4-Toluenesulfonyl chloride
4-Toluenesulfonyl chloride, also known as tosyl chloride or TsCl, is a chemical compound with the formula CH₃C₆H₄SO₂Cl. This organic compound is widely used in organic synthesis, primarily as a reagent for the introduction of the tosyl group into organic molecules. The tosyl group (Ts) serves as a protecting group for alcohols and amines, and as an activating group for nucleophilic substitution reactions.
Structure and Properties[edit]
4-Toluenesulfonyl chloride consists of a benzene ring substituted with a sulfonyl chloride group (SO₂Cl) and a methyl group (CH₃). The presence of the sulfonyl chloride group makes it a reactive compound, capable of forming esters and amides with alcohols and amines, respectively.
The compound is a white to slightly yellow crystalline solid with a pungent odor. It is poorly soluble in water but highly soluble in organic solvents such as acetone, benzene, and chloroform. The melting point of 4-toluenesulfonyl chloride is approximately 68-70°C, and it decomposes upon heating, emitting toxic fumes of sulfur oxides and hydrochloric acid.
Synthesis[edit]
4-Toluenesulfonyl chloride is synthesized from toluene through a two-step process. The first step involves the sulfonation of toluene with sulfuric acid to produce 4-toluenesulfonic acid. In the second step, 4-toluenesulfonic acid is treated with thionyl chloride (SOCl₂), resulting in the formation of 4-toluenesulfonyl chloride and the by-product sulfur dioxide (SO₂).
Applications[edit]
Protecting Group[edit]
One of the primary applications of 4-toluenesulfonyl chloride is as a protecting group for alcohols and amines. When reacted with an alcohol, it forms a tosylate ester, which is resistant to various nucleophilic and elimination reactions. This allows for selective reactions to be carried out on other parts of the molecule without affecting the protected functional group.
Nucleophilic Substitution[edit]
4-Toluenesulfonyl chloride is also used to activate halides for nucleophilic substitution reactions. The tosyl group can be introduced into a molecule in place of a less reactive leaving group, making the molecule more susceptible to attack by nucleophiles.
Other Applications[edit]
In addition to its use in organic synthesis, 4-toluenesulfonyl chloride is also employed in the preparation of dyes, pharmaceuticals, and agrochemicals. Its ability to introduce sulfonate esters and amides into molecules makes it a valuable tool in the synthesis of a wide range of organic compounds.
Safety and Handling[edit]
4-Toluenesulfonyl chloride is a corrosive substance and poses risks of burns upon contact with skin or eyes. It is also a respiratory irritant, and inhalation of its vapors can lead to respiratory distress. Proper personal protective equipment, including gloves, goggles, and a fume hood, should be used when handling this compound.
Environmental Impact[edit]
The production and use of 4-toluenesulfonyl chloride should be managed carefully to minimize its environmental impact. The compound is not readily biodegradable, and its release into the environment can pose risks to aquatic life. Efforts should be made to ensure that waste containing 4-toluenesulfonyl chloride is treated and disposed of in accordance with local regulations.
4-Toluenesulfonyl chloride gallery[edit]
-
P-Toluenesulfonyl chloride structure
-
Tosyl chloride 3D vdW