2-Hexanone
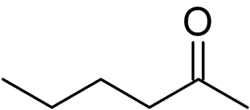
2-Hexanone (also known as Methyl n-butyl ketone, MBK, or propyl acetone) is an organic compound with the chemical formula C6H12O. It is a colorless liquid with a slightly sharp odor. It is produced on a large scale industrially, although in the home it can be found in certain paints and varnishes.
Chemical Properties[edit]
2-Hexanone is a ketone, which is a type of organic compound that contains a carbonyl group (C=O) bonded to two other carbon atoms. It is a six-carbon molecule, with the ketone functional group located at the second carbon atom, hence the '2' in its name. It is a polar molecule due to the presence of the carbonyl group, and it has a higher boiling point than similar sized hydrocarbons.
Production[edit]
2-Hexanone is produced industrially by the oxidation of hexane, a process that also yields other useful ketones. It can also be produced in the laboratory by the oxidation of 2-hexanol.
Uses[edit]
2-Hexanone is used as a solvent in some industrial applications, due to its ability to dissolve a wide range of organic compounds. It is also used in the production of adhesives and sealants.
Health Effects[edit]
Exposure to 2-hexanone can cause irritation to the eyes, skin, and respiratory tract. It can also cause neurological damage if exposure is prolonged or in high concentrations. It is considered a hazardous substance and should be handled with care.
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
