Hexane
Chemical compound
Hexane is a hydrocarbon with the chemical formula C6H14. It is an alkane with six carbon atoms and is a significant component of gasoline. Hexane is a colorless liquid at room temperature and is used in various industrial applications, particularly as a non-polar solvent.
Structure and Isomers[edit]
Hexane has several isomers, which are compounds with the same molecular formula but different structural arrangements. The linear form of hexane is known as n-hexane.
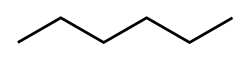
The isomers of hexane include:
- 2-Methylpentane

- 3-Methylpentane

- 2,3-Dimethylbutane

- 2,2-Dimethylbutane

These isomers differ in the branching of the carbon chain, which affects their physical properties such as boiling points and melting points.
Physical Properties[edit]
Hexane is a colorless liquid with a faint petroleum-like odor. It is highly volatile and has a boiling point of approximately 69 °C (156 °F). Hexane is insoluble in water but is miscible with many organic solvents such as ethanol, ether, and chloroform.
Production[edit]
Hexane is primarily obtained from the fractional distillation of crude oil. It is a major component of the alkane fraction in petroleum and is separated during the refining process. Industrially, hexane is produced by refining crude oil in a process that involves distillation and cracking.
Uses[edit]
Hexane is widely used as a solvent in the extraction of vegetable oils from seeds and as a cleaning agent in the textile, furniture, and leather industries. It is also used in the formulation of glues for shoes, roofing, and leather products.
In the laboratory, hexane is used as a solvent for chromatography and other chemical reactions due to its non-polar nature.
Health and Safety[edit]
Exposure to hexane can have adverse health effects. It is a neurotoxin and prolonged exposure can lead to peripheral neuropathy, characterized by numbness and weakness in the extremities. Hexane is also a volatile organic compound (VOC) and contributes to air pollution.
Proper safety measures, including adequate ventilation and protective equipment, are essential when handling hexane to minimize exposure risks.
Environmental Impact[edit]
Hexane is a volatile organic compound that can contribute to the formation of ground-level ozone, a component of smog. It is important to manage emissions of hexane to reduce its environmental impact.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian