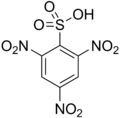
2,4,6-Trinitrobenzenesulfonic acid
2,4,6-Trinitrobenzenesulfonic acid[edit]
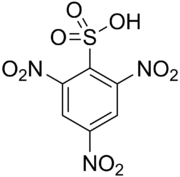
2,4,6-Trinitrobenzenesulfonic acid (TNBS) is a chemical compound that is commonly used in biochemical research. It is a derivative of benzenesulfonic acid with three nitro groups attached to the benzene ring.
Chemical Properties[edit]
2,4,6-Trinitrobenzenesulfonic acid is a sulfonic acid with the chemical formula C6H3N3O9S. It is a yellow crystalline solid that is soluble in water and other polar solvents. The presence of three nitro groups makes it a strong electrophile, which is useful in various chemical reactions.
Uses in Biochemistry[edit]
TNBS is widely used in the field of biochemistry for the detection and quantification of amino acids and proteins. It reacts with primary amines to form a colored complex, which can be measured spectrophotometrically. This property makes it useful in protein quantification assays.
Mechanism of Action[edit]
The reaction of TNBS with primary amines involves the formation of a Meisenheimer complex, where the nitro groups act as electron-withdrawing groups, facilitating the nucleophilic attack by the amine. This results in the formation of a stable colored product.
Safety and Handling[edit]
2,4,6-Trinitrobenzenesulfonic acid is a hazardous chemical and should be handled with care. It is an irritant to the skin, eyes, and respiratory tract. Proper safety precautions such as wearing gloves and goggles should be taken when handling this compound.
Related Compounds[edit]
Related Pages[edit]
-
2,4,6-Trinitrobenzenesulfonic_acid
-
2,4,6-Trinitrobenzenesulfonic_acid
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian