Benzenesulfonic acid
Benzenesulfonic acid is an organic compound with the chemical formula C6H6SO3H. It belongs to the class of compounds known as sulfonic acids, which are a group of organosulfur compounds characterized by a sulfur atom bonded to an oxygen atom and a hydroxyl group (-SO3H). Benzenesulfonic acid is a colorless, crystalline solid that is soluble in water and various organic solvents. It is an important industrial chemical used as a catalyst, in the production of detergents, dyes, and some pharmaceuticals, and as an intermediate in the synthesis of other chemical compounds.
Properties[edit]
Benzenesulfonic acid is a strong acid, stronger than comparable carboxylic acids. This strength is due to the stabilizing effect of the sulfonate group on the negative charge of the conjugate base, the benzenesulfonate ion. The sulfonate group is also responsible for the compound's high solubility in water. Benzenesulfonic acid has a melting point of approximately 40°C and decomposes before boiling.
Synthesis[edit]
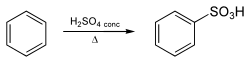
Benzenesulfonic acid is typically synthesized through the sulfonation of benzene using concentrated sulfuric acid or sulfur trioxide. The reaction is highly exothermic and produces benzenesulfonic acid along with heat and water:
C6H6 + H2SO4 → C6H5SO3H + H2O
Alternatively, benzenesulfonic acid can be produced by the hydrolysis of benzenesulfonyl chloride.
Applications[edit]
Benzenesulfonic acid and its derivatives are widely used in the chemical industry. They serve as intermediates in the synthesis of phenols, detergents, synthetic resins, and dyes. The acid's ability to act as a surfactant makes it valuable in the production of detergents and emulsifiers. It is also used as a catalyst in certain organic reactions, such as the alkylation of phenols and the hydration of nitriles to amides.
Safety[edit]
Benzenesulfonic acid is corrosive and can cause severe burns upon contact with skin or eyes. It should be handled with appropriate safety precautions, including the use of personal protective equipment such as gloves and safety glasses. In case of exposure, affected areas should be rinsed immediately with plenty of water, and medical advice should be sought.
Environmental Impact[edit]
As with many industrial chemicals, the release of benzenesulfonic acid into the environment can have detrimental effects. It is toxic to aquatic life and can cause long-term adverse effects in aquatic ecosystems. Therefore, waste containing benzenesulfonic acid should be treated and disposed of in accordance with local regulations to minimize its environmental impact.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
