(+)-cis-2-Aminomethylcyclopropane carboxylic acid
(+)-cis-2-Aminomethylcyclopropane carboxylic acid (also known as (+)-CAMP) is a chemical compound that is of interest in the field of pharmacology and neuroscience. It is a derivative of cyclopropane and is characterized by its unique three-membered ring structure.
Chemical Structure[edit]
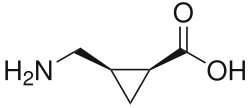
The chemical structure of (+)-cis-2-Aminomethylcyclopropane carboxylic acid consists of a cyclopropane ring with an aminomethyl group and a carboxylic acid group attached. The stereochemistry of the compound is denoted by the "cis" configuration, indicating that the substituents are on the same side of the cyclopropane ring.
3D Structure[edit]
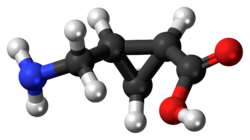
The three-dimensional structure of (+)-CAMP can be visualized using a ball-and-stick model, which highlights the spatial arrangement of atoms within the molecule. This model is useful for understanding the stereochemistry and potential interactions with biological targets.
Pharmacological Significance[edit]
(+)-CAMP is studied for its potential role as a neurotransmitter modulator. Its structure allows it to interact with receptors in the central nervous system, potentially influencing neurotransmission and synaptic plasticity. Research into compounds like (+)-CAMP is ongoing, with the aim of developing new therapeutic agents for neurological disorders.
Synthesis[edit]
The synthesis of (+)-cis-2-Aminomethylcyclopropane carboxylic acid involves several steps, starting from basic organic compounds. The process typically includes the formation of the cyclopropane ring, followed by the introduction of the aminomethyl and carboxylic acid groups. The stereochemistry is controlled through specific reaction conditions and catalysts.
Applications[edit]
While (+)-CAMP itself may not be used directly as a drug, its study provides valuable insights into the design of new pharmaceuticals. Understanding its interactions with biological systems can lead to the development of compounds with improved efficacy and safety profiles.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian