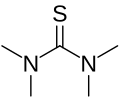
Tetramethylthiourea
Tetramethylthiourea is a chemical compound with the formula (CH3)4N2S. It is a white crystalline solid that is soluble in water and most organic solvents. Tetramethylthiourea is used as a building block in organic synthesis and as a reagent in the production of various pharmaceuticals.
Structure and Properties[edit]
Tetramethylthiourea is a thiourea derivative, which means it contains a thiourea functional group. This group consists of a sulfur atom bonded to two nitrogen atoms and two methyl groups. The sulfur atom in tetramethylthiourea is also bonded to two additional methyl groups, giving the compound its name.
The compound is a white crystalline solid at room temperature. It has a melting point of 104-106 °C and a boiling point of 280 °C. Tetramethylthiourea is soluble in water and most organic solvents, including ethanol, methanol, and acetone.
Synthesis[edit]
Tetramethylthiourea can be synthesized by the reaction of methylamine with carbon disulfide in the presence of a base. The reaction proceeds via the formation of a dithiocarbamate intermediate, which is then converted to tetramethylthiourea by reaction with additional methylamine.
Applications[edit]
Tetramethylthiourea is used as a building block in organic synthesis. It can act as a nucleophile, reacting with electrophiles to form new carbon-sulfur bonds. This makes it a useful reagent in the synthesis of various pharmaceuticals, including antibiotics and antiviral drugs.
In addition to its use in organic synthesis, tetramethylthiourea is also used as a corrosion inhibitor in the oil and gas industry. It forms a protective layer on the surface of metals, preventing them from reacting with oxygen and other corrosive substances.
Safety[edit]
Tetramethylthiourea is considered a hazardous substance. It can cause skin and eye irritation, and prolonged exposure can lead to respiratory problems. It is also harmful if swallowed. Therefore, it should be handled with care, using appropriate personal protective equipment.
See Also[edit]
-
Tetramethylthiourea
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian