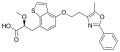
Aleglitazar
A dual PPAR agonist drug candidate
| Aleglitazar | |
|---|---|

| |
| INN | |
| Drug class | |
| Routes of administration | |
| Pregnancy category | |
| Bioavailability | |
| Metabolism | |
| Elimination half-life | |
| Excretion | |
| Legal status | |
| CAS Number | |
| PubChem | |
| DrugBank | |
| ChemSpider | |
| KEGG | |
Aleglitazar is a pharmaceutical compound that was developed as a dual peroxisome proliferator-activated receptor (PPAR) agonist. It was primarily investigated for its potential use in the treatment of type 2 diabetes mellitus and associated cardiovascular disease.
Mechanism of Action
Aleglitazar functions as a dual agonist of the PPAR_ and PPAR_ receptors. These receptors are nuclear hormone receptors that regulate the expression of genes involved in glucose and lipid metabolism. By activating both PPAR_ and PPAR_, aleglitazar was intended to improve insulin sensitivity, reduce blood glucose levels, and favorably modify lipid profiles.
PPAR_ Agonism
PPAR_ is primarily expressed in the liver, heart, kidney, and muscle tissues. Activation of PPAR_ leads to increased fatty acid oxidation, reduced triglyceride levels, and improved high-density lipoprotein (HDL) cholesterol levels. This action is beneficial in reducing atherosclerosis and cardiovascular risk.
PPAR_ Agonism
PPAR_ is predominantly found in adipose tissue, the colon, and the immune system. Activation of PPAR_ enhances insulin sensitivity, promotes adipocyte differentiation, and modulates inflammatory responses. This can lead to improved glycemic control in patients with type 2 diabetes.
Clinical Development
Aleglitazar was developed by F. Hoffmann-La Roche and underwent several clinical trials to assess its efficacy and safety in patients with type 2 diabetes and cardiovascular disease. Despite initial promising results, further studies revealed concerns regarding safety and adverse effects.
Adverse Effects
During clinical trials, aleglitazar was associated with several adverse effects, including edema, heart failure, and renal impairment. These safety concerns ultimately led to the discontinuation of its development.
Discontinuation
In 2013, the development of aleglitazar was halted after the results of the ALECARDIO trial indicated that the risks outweighed the potential benefits. The trial showed no significant reduction in cardiovascular events and highlighted the increased risk of adverse effects.
Related Pages
- Type 2 diabetes mellitus
- Cardiovascular disease
- Peroxisome proliferator-activated receptor
- Insulin resistance
Gallery
-
Chemical structure of Aleglitazar
Ad. Transform your life with W8MD's Budget GLP-1 injections from $49.99


W8MD offers a medical weight loss program to lose weight in Philadelphia. Our physician-supervised medical weight loss provides:
- Weight loss injections in NYC (generic and brand names):
- Zepbound / Mounjaro, Wegovy / Ozempic, Saxenda
- Most insurances accepted or discounted self-pay rates. We will obtain insurance prior authorizations if needed.
- Generic GLP1 weight loss injections from $49.99 for the starting dose of Semaglutide and $65.00 for Tirzepatide.
- Also offer prescription weight loss medications including Phentermine, Qsymia, Diethylpropion, Contrave etc.
NYC weight loss doctor appointmentsNYC weight loss doctor appointments
Start your NYC weight loss journey today at our NYC medical weight loss and Philadelphia medical weight loss clinics.
- Call 718-946-5500 to lose weight in NYC or for medical weight loss in Philadelphia 215-676-2334.
- Tags:NYC medical weight loss, Philadelphia lose weight Zepbound NYC, Budget GLP1 weight loss injections, Wegovy Philadelphia, Wegovy NYC, Philadelphia medical weight loss, Brookly weight loss and Wegovy NYC
|
WikiMD's Wellness Encyclopedia |
| Let Food Be Thy Medicine Medicine Thy Food - Hippocrates |
Medical Disclaimer: WikiMD is not a substitute for professional medical advice. The information on WikiMD is provided as an information resource only, may be incorrect, outdated or misleading, and is not to be used or relied on for any diagnostic or treatment purposes. Please consult your health care provider before making any healthcare decisions or for guidance about a specific medical condition. WikiMD expressly disclaims responsibility, and shall have no liability, for any damages, loss, injury, or liability whatsoever suffered as a result of your reliance on the information contained in this site. By visiting this site you agree to the foregoing terms and conditions, which may from time to time be changed or supplemented by WikiMD. If you do not agree to the foregoing terms and conditions, you should not enter or use this site. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian