Thiocarboxylic acid: Difference between revisions
CSV import |
CSV import |
||
| Line 28: | Line 28: | ||
{{Chemistry-stub}} | {{Chemistry-stub}} | ||
<gallery> | |||
File:Thioic_O-acid.svg|Thioic O-acid structure | |||
File:Thioic_acid.svg|Thioic acid structure | |||
</gallery> | |||
Latest revision as of 04:21, 18 February 2025
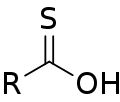
Thiocarboxylic acid is a type of organic compound characterized by the presence of a carboxylic acid group in which the oxygen atom is replaced by a sulfur atom. The general formula for thiocarboxylic acids is R-C(=S)-OH, where R represents an alkyl or aryl group. These compounds are part of a broader class of thioacids, which are sulfur analogs of oxygen-containing organic acids. Thiocarboxylic acids are of interest in both organic chemistry and biochemistry for their unique properties and potential applications.
Properties and Synthesis[edit]
Thiocarboxylic acids exhibit distinct properties due to the presence of the sulfur atom. The sulfur atom is larger and less electronegative than oxygen, which affects the acidity and reactivity of these compounds. Thiocarboxylic acids are generally weaker acids than their carboxylic acid counterparts. They are also more nucleophilic, making them useful in various synthetic applications.
The synthesis of thiocarboxylic acids can be achieved through several methods. One common approach involves the thionation of carboxylic acids or their derivatives using reagents such as phosphorus pentasulfide (P2S5) or Lawesson's reagent. Another method is the direct sulfurization of acyl chlorides with hydrogen sulfide (H2S) or thiourea.
Applications[edit]
Thiocarboxylic acids and their derivatives find applications in various fields. In organic synthesis, they are used as building blocks for the preparation of a wide range of sulfur-containing compounds, including thioesters, thioamides, and dithiocarbamates. These compounds are important in the synthesis of pharmaceuticals, agrochemicals, and polymers.
In biochemistry, thiocarboxylic acids are of interest due to their potential as inhibitors of certain enzymes. For example, they can act as inhibitors of acetylcholinesterase, an enzyme involved in the breakdown of the neurotransmitter acetylcholine. This property makes them potential candidates for the treatment of diseases such as Alzheimer's disease.
Safety and Environmental Considerations[edit]
Like many sulfur-containing compounds, thiocarboxylic acids can have unpleasant odors and may pose certain health and environmental risks. Handling of these compounds should be done with appropriate safety precautions, including the use of fume hoods and personal protective equipment. Disposal of thiocarboxylic acids and their derivatives should follow regulations to minimize environmental impact.
See Also[edit]
References[edit]
<references/>
-
Thioic O-acid structure
-
Thioic acid structure