Lumateperone: Difference between revisions
CSV import |
CSV import Tags: mobile edit mobile web edit |
||
| Line 5: | Line 5: | ||
| image = ITI-007.svg | | image = ITI-007.svg | ||
| image2 = | | image2 = | ||
| width = | | width = 200 | ||
| alt = | |||
| caption = Chemical structure of Lumateperone | |||
}} | }} | ||
| Line 11: | Line 13: | ||
==Pharmacology== | ==Pharmacology== | ||
Lumateperone acts as a [[serotonin receptor]] antagonist, [[dopamine receptor]] | Lumateperone acts as a [[serotonin receptor]] antagonist, [[dopamine receptor]] partial agonist, and [[serotonin transporter]] inhibitor. Its mechanism of action is thought to involve modulation of the [[serotonin]] (5-HT2A) and [[dopamine]] (D2) receptors, which are implicated in the pathophysiology of schizophrenia and bipolar disorder. This unique receptor activity profile is believed to contribute to its efficacy and tolerability. | ||
===Mechanism of Action=== | ===Mechanism of Action=== | ||
Lumateperone's antipsychotic effects are | Lumateperone's antipsychotic effects are primarily attributed to its antagonism of the 5-HT2A receptor and partial agonism at the D2 receptor. Additionally, it has moderate affinity for the [[serotonin transporter]], which may enhance its therapeutic effects. Unlike many other antipsychotics, lumateperone has minimal activity at the [[histamine]] and [[muscarinic acetylcholine receptors]], which may reduce the risk of side effects such as sedation and anticholinergic effects. | ||
==Clinical Use== | ==Clinical Use== | ||
Lumateperone is indicated for the treatment of schizophrenia in adults. It is also approved for the treatment of depressive episodes associated with bipolar I or II disorder | Lumateperone is indicated for the treatment of schizophrenia in adults. It is also approved for the treatment of depressive episodes associated with bipolar I or II disorder, either as monotherapy or as adjunctive therapy with [[lithium]] or [[valproate]]. | ||
===Dosage and Administration=== | ===Dosage and Administration=== | ||
Lumateperone is administered orally, typically once daily with | Lumateperone is administered orally, typically at a dose of 42 mg once daily with food. The dosage may be adjusted based on clinical response and tolerability. | ||
==Side Effects== | ==Side Effects== | ||
Common side effects of lumateperone include somnolence, dry mouth, dizziness | Common side effects of lumateperone include [[somnolence]], [[dry mouth]], and [[dizziness]]. It has a lower risk of causing [[extrapyramidal symptoms]] compared to other antipsychotics. Weight gain and metabolic disturbances are also less common with lumateperone. | ||
== | ==History== | ||
Lumateperone | Lumateperone was developed by Intra-Cellular Therapies, a biopharmaceutical company focused on the development of novel drugs for the treatment of neuropsychiatric and neurological disorders. The drug was approved by the FDA for the treatment of schizophrenia in December 2019 and for bipolar depression in December 2021. | ||
== | ==Research== | ||
Ongoing research is exploring the potential of lumateperone in other psychiatric conditions, including [[major depressive disorder]] and [[autism spectrum disorder]]. Studies are also investigating its long-term safety and efficacy. | |||
==Related pages== | ==Related pages== | ||
| Line 38: | Line 37: | ||
* [[Bipolar disorder]] | * [[Bipolar disorder]] | ||
* [[Atypical antipsychotic]] | * [[Atypical antipsychotic]] | ||
* [[ | * [[Serotonin]] | ||
* [[ | * [[Dopamine]] | ||
[[Category:Atypical antipsychotics]] | [[Category:Atypical antipsychotics]] | ||
[[Category:Serotonin antagonists]] | [[Category:Serotonin receptor antagonists]] | ||
[[Category:Dopamine | [[Category:Dopamine receptor modulators]] | ||
Latest revision as of 01:16, 6 March 2025
Atypical antipsychotic medication
| Lumateperone | |
|---|---|
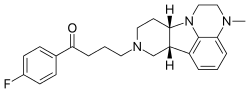
| |
| INN | |
| Drug class | |
| Routes of administration | |
| Pregnancy category | |
| Bioavailability | |
| Metabolism | |
| Elimination half-life | |
| Excretion | |
| Legal status | |
| CAS Number | |
| PubChem | |
| DrugBank | |
| ChemSpider | |
| KEGG | |
Lumateperone, marketed under the brand name Caplyta, is an atypical antipsychotic medication used primarily in the treatment of schizophrenia and bipolar disorder. It was developed by Intra-Cellular Therapies and received approval from the United States Food and Drug Administration (FDA) in December 2019.
Pharmacology[edit]
Lumateperone acts as a serotonin receptor antagonist, dopamine receptor partial agonist, and serotonin transporter inhibitor. Its mechanism of action is thought to involve modulation of the serotonin (5-HT2A) and dopamine (D2) receptors, which are implicated in the pathophysiology of schizophrenia and bipolar disorder. This unique receptor activity profile is believed to contribute to its efficacy and tolerability.
Mechanism of Action[edit]
Lumateperone's antipsychotic effects are primarily attributed to its antagonism of the 5-HT2A receptor and partial agonism at the D2 receptor. Additionally, it has moderate affinity for the serotonin transporter, which may enhance its therapeutic effects. Unlike many other antipsychotics, lumateperone has minimal activity at the histamine and muscarinic acetylcholine receptors, which may reduce the risk of side effects such as sedation and anticholinergic effects.
Clinical Use[edit]
Lumateperone is indicated for the treatment of schizophrenia in adults. It is also approved for the treatment of depressive episodes associated with bipolar I or II disorder, either as monotherapy or as adjunctive therapy with lithium or valproate.
Dosage and Administration[edit]
Lumateperone is administered orally, typically at a dose of 42 mg once daily with food. The dosage may be adjusted based on clinical response and tolerability.
Side Effects[edit]
Common side effects of lumateperone include somnolence, dry mouth, and dizziness. It has a lower risk of causing extrapyramidal symptoms compared to other antipsychotics. Weight gain and metabolic disturbances are also less common with lumateperone.
History[edit]
Lumateperone was developed by Intra-Cellular Therapies, a biopharmaceutical company focused on the development of novel drugs for the treatment of neuropsychiatric and neurological disorders. The drug was approved by the FDA for the treatment of schizophrenia in December 2019 and for bipolar depression in December 2021.
Research[edit]
Ongoing research is exploring the potential of lumateperone in other psychiatric conditions, including major depressive disorder and autism spectrum disorder. Studies are also investigating its long-term safety and efficacy.