Thioacetazone: Difference between revisions
CSV import |
CSV import |
||
| Line 1: | Line 1: | ||
{{Short description|Antitubercular medication}} | |||
{{Drugbox | |||
| verifiedrevid = 477241679 | |||
| image = Thioacetazone.svg | |||
| image2 = Thioacetazone_3D_spacefill.png | |||
| width = 200 | |||
| alt = Thioacetazone.svg | |||
| alt2 = Thioacetazone_3D_spacefill.png | |||
}} | |||
'''Thioacetazone''' is an [[antibacterial]] medication primarily used in the treatment of [[tuberculosis]] (TB). It is known for its role in combination therapy for [[Mycobacterium tuberculosis]], the bacterium responsible for TB. Thioacetazone is particularly noted for its use in resource-limited settings due to its cost-effectiveness. | |||
Thioacetazone | |||
== | ==Chemical Structure== | ||
Thioacetazone | [[File:Thioacetazone.svg|thumb|right|Chemical structure of Thioacetazone]] | ||
Thioacetazone is a thiosemicarbazone derivative, characterized by its sulfur-containing moiety. The chemical structure of thioacetazone includes a thiosemicarbazone group attached to an aromatic ring, which is crucial for its antibacterial activity. | |||
== | ==Mechanism of Action== | ||
Thioacetazone works by inhibiting the synthesis of mycolic acids, essential components of the [[mycobacterial cell wall]]. This inhibition disrupts the integrity of the cell wall, leading to the death of the bacterium. The drug is bacteriostatic, meaning it inhibits the growth and reproduction of bacteria rather than killing them directly. | |||
== | ==Clinical Use== | ||
Thioacetazone is used in combination with other antitubercular drugs such as [[isoniazid]] and [[rifampicin]]. It is particularly useful in the treatment of drug-resistant TB strains. However, its use has declined in many parts of the world due to the availability of more effective and less toxic alternatives. | |||
== | ==Side Effects== | ||
The use of thioacetazone is associated with several side effects, the most significant being [[skin reactions]] such as [[Stevens-Johnson syndrome]] and [[toxic epidermal necrolysis]]. These severe skin reactions are more common in patients with [[HIV/AIDS]], which has led to caution in its use among these populations. | |||
==History== | |||
Thioacetazone was first introduced in the 1940s as a treatment for tuberculosis. It gained popularity due to its low cost and effectiveness in combination therapy. However, the emergence of severe side effects and the development of more effective drugs have limited its use in modern TB treatment regimens. | |||
==Related Pages== | |||
* [[Tuberculosis treatment]] | * [[Tuberculosis treatment]] | ||
* [[ | * [[Antitubercular agents]] | ||
* [[ | * [[Mycobacterium tuberculosis]] | ||
==See Also== | |||
[[ | * [[Isoniazid]] | ||
[[ | * [[Rifampicin]] | ||
[[ | * [[Ethambutol]] | ||
[[Category:Antitubercular agents]] | |||
[[Category:Thiosemicarbazones]] | |||
[[Category:World Health Organization essential medicines]] | |||
Latest revision as of 18:52, 23 March 2025
Antitubercular medication
| Thioacetazone | |
|---|---|
| |
| INN | |
| Drug class | |
| Routes of administration | |
| Pregnancy category | |
| Bioavailability | |
| Metabolism | |
| Elimination half-life | |
| Excretion | |
| Legal status | |
| CAS Number | |
| PubChem | |
| DrugBank | |
| ChemSpider | |
| KEGG | |
Thioacetazone is an antibacterial medication primarily used in the treatment of tuberculosis (TB). It is known for its role in combination therapy for Mycobacterium tuberculosis, the bacterium responsible for TB. Thioacetazone is particularly noted for its use in resource-limited settings due to its cost-effectiveness.
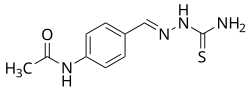
Chemical Structure[edit]

Thioacetazone is a thiosemicarbazone derivative, characterized by its sulfur-containing moiety. The chemical structure of thioacetazone includes a thiosemicarbazone group attached to an aromatic ring, which is crucial for its antibacterial activity.
Mechanism of Action[edit]
Thioacetazone works by inhibiting the synthesis of mycolic acids, essential components of the mycobacterial cell wall. This inhibition disrupts the integrity of the cell wall, leading to the death of the bacterium. The drug is bacteriostatic, meaning it inhibits the growth and reproduction of bacteria rather than killing them directly.
Clinical Use[edit]
Thioacetazone is used in combination with other antitubercular drugs such as isoniazid and rifampicin. It is particularly useful in the treatment of drug-resistant TB strains. However, its use has declined in many parts of the world due to the availability of more effective and less toxic alternatives.
Side Effects[edit]
The use of thioacetazone is associated with several side effects, the most significant being skin reactions such as Stevens-Johnson syndrome and toxic epidermal necrolysis. These severe skin reactions are more common in patients with HIV/AIDS, which has led to caution in its use among these populations.
History[edit]
Thioacetazone was first introduced in the 1940s as a treatment for tuberculosis. It gained popularity due to its low cost and effectiveness in combination therapy. However, the emergence of severe side effects and the development of more effective drugs have limited its use in modern TB treatment regimens.