Viltolarsen
An article about the drug Viltolarsen
| Viltolarsen | |
|---|---|
|
|
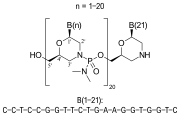
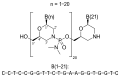
Viltolarsen is a pharmaceutical drug used in the treatment of Duchenne muscular dystrophy (DMD), a severe type of muscular dystrophy characterized by rapid progression of muscle degeneration. Viltolarsen is specifically designed to target mutations in the dystrophin gene that are amenable to exon 53 skipping.
Mechanism of Action[edit]
Viltolarsen is an antisense oligonucleotide that works by binding to exon 53 of the dystrophin pre-mRNA. This binding causes the exon to be skipped during mRNA splicing, resulting in the production of a truncated but functional dystrophin protein. This mechanism is intended to partially restore the function of the dystrophin protein in patients with specific genetic mutations.
Clinical Use[edit]
Viltolarsen is indicated for the treatment of patients with Duchenne muscular dystrophy who have a confirmed mutation of the dystrophin gene that is amenable to exon 53 skipping. It is administered via intravenous infusion and is typically given on a weekly basis.
Side Effects[edit]
Common side effects of Viltolarsen include injection site reactions, upper respiratory tract infections, and fever. Patients receiving Viltolarsen should be monitored for potential adverse reactions, and the drug should be used with caution in patients with renal impairment.
Development and Approval[edit]
Viltolarsen was developed by Nippon Shinyaku and NS Pharma. It received accelerated approval from the United States Food and Drug Administration (FDA) in 2020 based on its ability to increase dystrophin production in skeletal muscle. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
Related pages[edit]
-
Viltolarsen
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian