Trisodium citrate
Trisodium citrate[edit]
Trisodium citrate, also known as sodium citrate, is the sodium salt of citric acid. It possesses the chemical formula Na_C_H_O_ and is commonly used in the food and pharmaceutical industries. Trisodium citrate is a white, crystalline powder that is highly soluble in water and has a slightly salty, tart flavor.
Chemical Properties[edit]
Trisodium citrate is derived from citric acid, a weak organic acid that is naturally found in citrus fruits. The compound is formed by neutralizing citric acid with sodium hydroxide or sodium carbonate. In aqueous solution, trisodium citrate dissociates into three sodium ions and one citrate ion.
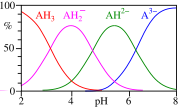
Speciation[edit]
The speciation of citric acid in solution is an important aspect of understanding the behavior of trisodium citrate. As shown in the diagram, citric acid can exist in multiple forms depending on the pH of the solution. At higher pH levels, the citrate ion predominates, which is the form present in trisodium citrate solutions.
Uses[edit]
Trisodium citrate is widely used in the food industry as an emulsifier, preservative, and flavoring agent. It is commonly found in beverages, dairy products, and processed foods. In the pharmaceutical industry, trisodium citrate is used as an anticoagulant in blood collection tubes and as a buffering agent in medications.
Food Industry[edit]
In the food industry, trisodium citrate acts as a pH buffer and helps maintain the stability of emulsions. It is often used in carbonated beverages to enhance flavor and in cheese production to improve texture.
Medical Applications[edit]
In medicine, trisodium citrate is used to prevent blood clotting during blood transfusions and in dialysis treatments. It is also used in oral rehydration solutions to treat dehydration.
Safety[edit]
Trisodium citrate is generally recognized as safe (GRAS) by the Food and Drug Administration (FDA) when used in accordance with good manufacturing practices. However, excessive consumption may lead to gastrointestinal discomfort.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian