Triphenylmethane
Triphenylmethane[edit]
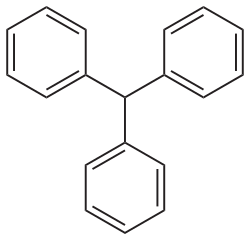
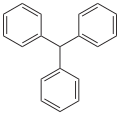
Triphenylmethane is an organic compound with the formula C__H__. It is a colorless solid, although commercial samples can appear yellowish. Triphenylmethane is the parent compound of many dyes, known as triphenylmethane dyes, which are used in a variety of applications.
Structure and Properties[edit]
Triphenylmethane consists of a central carbon atom bonded to three phenyl groups. The compound is a derivative of methane, where three hydrogen atoms are replaced by phenyl groups. This structure imparts significant stability to the molecule due to the resonance of the phenyl rings.
The compound is relatively nonpolar and insoluble in water but soluble in organic solvents such as benzene and chloroform.
Synthesis[edit]
Triphenylmethane can be synthesized by the Friedel-Crafts alkylation of benzene with chloroform in the presence of a Lewis acid such as aluminum chloride. This reaction involves the formation of a carbocation intermediate, which is stabilized by the phenyl groups.
Applications[edit]
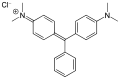
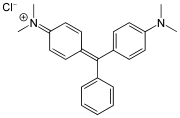
While triphenylmethane itself is not widely used, its derivatives are important in the production of dyes. These dyes, known as triphenylmethane dyes, include compounds such as malachite green and bromocresol green.
Triphenylmethane Dyes[edit]
Triphenylmethane dyes are characterized by their brilliant colors and are used in textiles, inks, and as biological stains. They are synthesized by introducing various functional groups into the triphenylmethane structure, which alters the electronic properties and color of the dye.
Environmental and Health Aspects[edit]
Some triphenylmethane dyes have been found to be toxic and can pose environmental hazards. For example, malachite green is known to be a potential carcinogen and is regulated in many countries.
Related Pages[edit]
References[edit]
-
Triphenylmethane structure
-
Triphenylmethane anion
-
Malachite green structure
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian