Trimethylamine
Trimethylamine[edit]
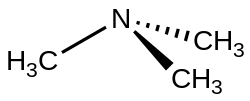
Trimethylamine (TMA) is an organic compound with the formula (CH_)_N. It is a tertiary amine and is a colorless, hygroscopic, and flammable liquid at room temperature. Trimethylamine is known for its strong "fishy" odor, which is detectable at very low concentrations.
Structure and Properties[edit]
Trimethylamine is a pyramidal molecule, as depicted in the structural formula. The nitrogen atom in trimethylamine is sp_ hybridized, resulting in a trigonal pyramidal geometry. This geometry is typical for amines, where the lone pair of electrons on the nitrogen atom contributes to the shape.
Trimethylamine is a polar molecule due to the presence of the lone pair on the nitrogen, which creates a dipole moment. It is soluble in water and many organic solvents, and it can form hydrogen bonds with water molecules.
Occurrence[edit]
Trimethylamine is naturally occurring and is found in various biological systems. It is produced by the decomposition of organic matter, particularly fish, which is why it is often associated with the smell of rotting fish. In humans, trimethylamine is produced in the gut during the digestion of certain foods and is metabolized by the liver.
Biological Role[edit]
In the human body, trimethylamine is converted to trimethylamine N-oxide (TMAO) by the enzyme flavin-containing monooxygenase 3 (FMO3). TMAO is excreted in the urine. A genetic disorder known as trimethylaminuria, or "fish odor syndrome," occurs when there is a deficiency in FMO3, leading to the accumulation of trimethylamine and a characteristic body odor.
Industrial Uses[edit]
Trimethylamine is used in the production of various chemicals and pharmaceuticals. It is a precursor to choline, an essential nutrient, and is used in the synthesis of quaternary ammonium compounds, which are used as disinfectants and antistatic agents.
Safety and Handling[edit]
Trimethylamine is a flammable liquid and should be handled with care. It can cause irritation to the skin, eyes, and respiratory tract. Proper personal protective equipment (PPE) should be worn when handling trimethylamine, and it should be stored in a well-ventilated area away from sources of ignition.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian