Trimethyl orthoformate
Trimethyl orthoformate (TMOF) is an organic compound with the formula C4H10O3. It is a colorless liquid that is used in organic synthesis as a reagent for the introduction of the methoxy group.
Structure and properties[edit]
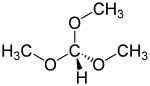
Trimethyl orthoformate is the simplest orthoester, consisting of a central carbon atom bonded to three methoxy groups. The presence of these methoxy groups makes it a useful reagent in chemical reactions where the introduction of a methoxy group is desired.
Synthesis[edit]
Trimethyl orthoformate can be synthesized by the reaction of methanol with hydrogen cyanide followed by hydrolysis. Alternatively, it can be prepared by the reaction of methyl formate with methanol in the presence of an acid catalyst.
Applications[edit]
In organic chemistry, trimethyl orthoformate is used as a protecting group for alcohols and carboxylic acids. It is also employed in the Pinner reaction to form esters from nitriles.
Protecting group[edit]
As a protecting group, trimethyl orthoformate can be used to protect hydroxyl groups by converting them into orthoesters. This protection is useful in multi-step synthesis where the hydroxyl group needs to be preserved from unwanted reactions.
Pinner reaction[edit]
In the Pinner reaction, trimethyl orthoformate reacts with nitriles in the presence of an acid to form imidates, which can be hydrolyzed to esters. This reaction is valuable in the synthesis of esters from nitriles.
Safety and handling[edit]
Trimethyl orthoformate is flammable and should be handled with care. It should be stored in a cool, dry place away from sources of ignition. Proper personal protective equipment should be worn when handling this chemical.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian