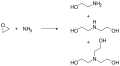
Triethanolamine
A chemical compound used in various industrial applications
Triethanolamine (TEA) is a viscous organic compound that is both a tertiary amine and a triol. A triol is a molecule with three alcohol groups. Like other amines, triethanolamine is a weak base.
Structure and properties[edit]
Triethanolamine is a colorless, viscous liquid. It is hygroscopic and has a slight ammonia-like odor. The chemical formula for triethanolamine is C6H15NO3, and its molecular structure consists of a central nitrogen atom bonded to three ethanol groups. This structure allows it to act as both a base and an alcohol.
Synthesis[edit]
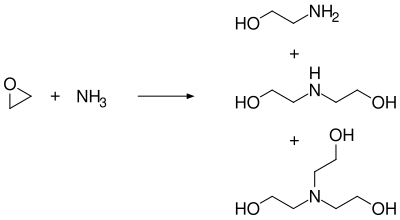
Triethanolamine is produced by the reaction of ethylene oxide with ammonia. The reaction can be controlled to produce monoethanolamine, diethanolamine, or triethanolamine, depending on the stoichiometry and reaction conditions.
Applications[edit]
Triethanolamine is used in a variety of industrial and consumer products. It is commonly used as a surfactant and emulsifier in cosmetics and personal care products, such as lotions, creams, and shampoos. In the textile industry, it is used as a softening agent. It also serves as a corrosion inhibitor in metalworking fluids and as a neutralizing agent in detergents.
Safety and environmental impact[edit]
Triethanolamine is considered to be of low toxicity, but it can cause skin and eye irritation. Prolonged exposure may lead to more serious health effects. It is important to handle it with appropriate safety measures, such as wearing protective clothing and eye protection.
Related pages[edit]
-
Triethanolamine
-
Reaction of ethylene oxide with ammonia
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian