Tolidine
Tolidine[edit]
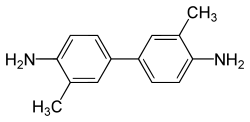
Tolidine refers to a group of chemical compounds that are derivatives of benzidine. These compounds are primarily used in the production of dyes and pigments. Tolidine is known for its application in the synthesis of azo dyes, which are used in various industries, including textiles and printing.
Chemical Structure[edit]
Tolidine compounds are characterized by their aromatic structure, which includes two amine groups. The most common form of tolidine is 2-tolidine, which has the chemical formula C14H16N2. The structure consists of two benzene rings connected by a diazo linkage, with each benzene ring bearing a methyl group and an amine group.
Synthesis[edit]
Tolidine is synthesized through a series of chemical reactions starting from nitrobenzene. The process involves the reduction of nitrobenzene to aniline, followed by diazotization and coupling reactions to form the tolidine structure. This synthesis is crucial for producing tolidine on an industrial scale.
Applications[edit]
Tolidine is primarily used in the manufacture of dyes and pigments. It serves as an intermediate in the production of azo dyes, which are known for their vibrant colors and stability. These dyes are widely used in the textile industry for coloring fabrics and in the printing industry for inks.
Safety and Environmental Concerns[edit]
Like many aromatic amines, tolidine is associated with potential health risks. It is considered a carcinogen, and exposure to tolidine should be minimized. Proper handling and disposal procedures are essential to prevent environmental contamination and protect human health.
Related Compounds[edit]
Tolidine is related to other aromatic amines such as benzidine and o-tolidine. These compounds share similar chemical properties and applications but differ in their specific structures and uses.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian