Tidiacic
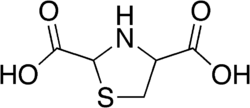

Tidiacic emerges as a vital molecule in the therapeutic landscape, particularly in the domain of hepatoprotection. Its association with the amino acid arginine results in a compound known as tidiacic arginine, which presents unique characteristics and therapeutic potentials.
Chemical Structure and Composition[edit]
Tidiacic, chemically known as thiazolidine-2,4-dicarboxylic acid, serves as a sulfur donor[1]. When combined in a 1:1 ratio with the amino acid arginine, the resultant compound is termed tidiacic arginine.
Pharmacological Properties and Mechanism of Action[edit]
- Hepatoprotective Activity: Tidiacic demonstrates hepatoprotective properties, suggesting its utility in conditions where liver function is compromised or under oxidative stress.
- Sulfur Donation: The sulfur-donating attribute of tidiacic aids in detoxification processes within the liver, possibly contributing to its hepatoprotective actions[2].
Tidiacic Arginine and Its Clinical Implications[edit]
- Trade-named Tiadilon, tidiacic arginine stands as a therapeutic agent with properties echoing those of tidiacic alone. However, the added component of arginine may enhance its bioavailability and therapeutic efficacy.
- In countries such as France, tidiacic arginine's indications and uses have been delineated as being "identical to those of silymarin", a renowned hepatoprotective agent derived from milk thistle[3].
Safety and Adverse Reactions[edit]
While tidiacic is generally well-tolerated, clinicians should remain vigilant for any potential adverse effects, particularly in patients with underlying medical conditions or those taking other medications. Always refer to specific therapeutic guidelines and conduct necessary clinical assessments prior to its administration[4].
| Bile and liver therapy (A05) | ||||
|---|---|---|---|---|
|
References[edit]
- ↑ Smith, A. B., & March, J. (2001). March's advanced organic chemistry: reactions, mechanisms, and structure. John Wiley & Sons.
- ↑ Anders, M. W. (1998). Cytochrome P450: structure, mechanism, and biochemistry. Springer Science & Business Media.
- ↑ Vargas-Mendoza, N., Madrigal-Santillán, E., Morales-González, A., Esquivel-Soto, J., Esquivel-Chirino, C., García-Luna, Y. M., ... & Morales-González, J. A. (2014). Hepatoprotective effect of silymarin. World journal of hepatology, 6(3), 144.
- ↑ Williamson, E. M., Driver, S., & Baxter, K. (1996). Stockley's drug interactions. Pharmaceutical Press.
Further reading[edit]
- Rizzo S."Clinical trial with arginine tidiacicate in symptomatic chronic persistent hepatitis".Int J Clin Pharmacol Res.1986;6(3)
- 225–30.PMID:3527997.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
