Myosin
Myosin is a protein that plays a crucial role in muscle contraction and a wide range of other cellular processes. It is a motor protein that converts chemical energy in the form of adenosine triphosphate (ATP) into mechanical energy, thereby generating force and movement. Myosins are a large family of proteins, with over 20 classes identified in both eukaryotic and prokaryotic cells, each serving distinct functions in cellular activities including muscle contraction, cell division, and vesicle transport.
Structure and Function[edit]
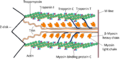
Myosin molecules are composed of two major parts: a head and a tail. The head domain binds to actin filaments and hydrolyzes ATP to generate force, while the tail domain determines the specific function of the myosin by directing its binding to different cellular components.
Myosin and Muscle Contraction[edit]
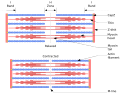
In muscle cells, myosin II plays a pivotal role in muscle contraction through the sliding filament theory. This process involves myosin head groups attaching to actin filaments and pulling them past one another, shortening the muscle cell. This action is powered by the hydrolysis of ATP by the myosin head, which undergoes a conformational change, generating the force required for movement.
Other Cellular Functions[edit]
Beyond muscle contraction, myosins are involved in various cellular functions. For example, myosin V is critical for the transport of organelles and vesicles within cells, playing a key role in endocytosis and exocytosis. Myosin XI is known to accelerate cytoplasmic streaming in plant cells, enhancing the distribution of nutrients and other molecules within the cell.
Types of Myosin[edit]
The myosin superfamily is divided into several classes, with myosin II being the most well-studied due to its role in muscle contraction. Other classes, such as myosin V, VI, and XI, have been identified in various organisms and cell types, each with unique functions and structural characteristics.
Regulation[edit]
The activity of myosin is tightly regulated by calcium ions and phosphorylation. In muscle cells, the binding of calcium to the troponin complex on actin filaments exposes myosin-binding sites, allowing for muscle contraction. Phosphorylation of myosin light chains can also modulate myosin activity, affecting cellular movement and muscle contraction.
Clinical Significance[edit]
Mutations in myosin genes can lead to a variety of genetic disorders, including certain forms of cardiomyopathy, a disease of the heart muscle, and hearing loss, due to the role of specific myosins in the inner ear. Understanding the function and regulation of myosin is crucial for developing treatments for these and other myosin-related diseases.
Research and Future Directions[edit]
Ongoing research aims to further elucidate the diverse roles of myosin proteins in cellular processes and disease. The development of drugs targeting myosin activity presents a promising avenue for treating myosin-related disorders, including heart disease and certain types of cancer.
|
This article is a stub You can help WikiMD by registering and expanding it with useful details, internal links, formatting, and categories. Editing is available only to registered and verified users. WikiMD is a comprehensive, free health and wellness encyclopedia. |
-
Myosine
-
Myosin Unrooted Tree
-
Sarcomere
-
Cardiac sarcomere structure
-
Myosin II AU to rel
-
Myosin V
-
Image animated
-
Myosin VI 2V26
-
Querbrückenzyklus 1
-
Querbrückenzyklus 2
-
Querbrückenzyklus 3
-
Querbrückenzyklus 4
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian