Tert-Butyllithium


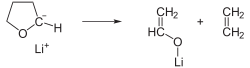
Tert-Butyllithium (t-BuLi) is a highly reactive organolithium reagent with the chemical formula (CH₃)₃CLi. It is commonly used in organic synthesis for the deprotonation of weak acids and as a strong base in various chemical reactions.
Properties[edit]
Tert-Butyllithium is a colorless liquid that is highly flammable and pyrophoric, meaning it can ignite spontaneously upon exposure to air. It is typically handled in a hydrocarbon solvent such as hexane or pentane to mitigate its reactivity. The compound is known for its strong basicity and nucleophilicity, making it a valuable reagent in synthetic chemistry.
Synthesis[edit]
Tert-Butyllithium is synthesized by the reaction of tert-butyl chloride with lithium metal in the presence of a hydrocarbon solvent. The reaction proceeds as follows:
(CH₃)₃CCl + 2Li → (CH₃)₃CLi + LiCl
This process requires careful control of temperature and exclusion of moisture and air to prevent unwanted side reactions.
Applications[edit]
Tert-Butyllithium is widely used in organic chemistry for various applications, including:
- **Deprotonation Reactions**: It is used to deprotonate weak acids, such as alcohols, amines, and alkynes, to generate the corresponding carbanions.
- **Metalation Reactions**: t-BuLi is employed to introduce lithium into organic molecules, a process known as metalation.
- **Polymerization**: It is used as an initiator in the anionic polymerization of certain monomers, such as styrene and butadiene.
Safety and Handling[edit]
Due to its extreme reactivity, tert-butyllithium must be handled with great care. It is typically stored under an inert atmosphere, such as nitrogen or argon, to prevent contact with air and moisture. Proper personal protective equipment (PPE), including gloves and eye protection, is essential when working with this reagent.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
