Tazomeline
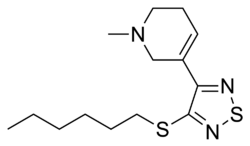
Tazomeline is a pharmacological agent that was developed by Eli Lilly and Company. It is classified as a M1 muscarinic receptor agonist. Tazomeline was primarily investigated for its potential use in the treatment of Alzheimer's disease and schizophrenia, but development was discontinued in the late 1990s.
History[edit]
Tazomeline was first synthesized by researchers at Eli Lilly and Company in the early 1990s. The compound was developed as part of a larger effort to discover new treatments for neurodegenerative diseases and psychiatric disorders. Initial studies showed that Tazomeline had a high affinity for the M1 muscarinic receptor, which is involved in many important cognitive functions.
Pharmacology[edit]
Tazomeline is a selective agonist for the M1 muscarinic receptor. This receptor is one of five subtypes of muscarinic receptors (M1-M5) and is primarily found in the brain and gastric glands. Activation of the M1 receptor by Tazomeline can enhance cognitive function and potentially alleviate symptoms of diseases like Alzheimer's and schizophrenia.
Clinical Trials[edit]
Several clinical trials were conducted to assess the safety and efficacy of Tazomeline in the 1990s. These trials focused on patients with Alzheimer's disease and schizophrenia. However, the results were mixed, and development of Tazomeline was ultimately discontinued.
Discontinuation[edit]
The development of Tazomeline was discontinued in the late 1990s. The reasons for this decision are not publicly available, but it is likely that the compound did not meet the necessary safety and efficacy standards in clinical trials.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
