Pyrone

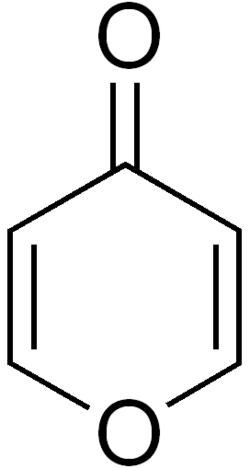
Pyrone is an organic compound with the molecular formula C₅H₄O₂. It is a heterocyclic compound that contains a six-membered ring with two double bonds and an oxygen atom. Pyrones are divided into two isomers: α-pyrone and γ-pyrone, which differ in the position of the carbonyl group.
Structure and Isomers[edit]
Pyrones are characterized by a six-membered ring structure containing one oxygen atom and two double bonds. The two main isomers of pyrone are:
- α-Pyrone (2-pyrone): The carbonyl group is located at the second position of the ring.
- γ-Pyrone (4-pyrone): The carbonyl group is located at the fourth position of the ring.
Synthesis[edit]
Pyrones can be synthesized through various chemical reactions. One common method involves the cyclization of β-keto esters in the presence of an acid catalyst. Another method includes the Perkin reaction, which involves the condensation of an aromatic aldehyde with an anhydride.
Reactivity[edit]
Pyrones exhibit unique reactivity due to the presence of the carbonyl group and the conjugated double bonds. They can undergo various chemical reactions, including:
- Diels-Alder reaction: Pyrones can act as dienes in Diels-Alder reactions, forming cyclohexene derivatives.
- Nucleophilic addition: The carbonyl group in pyrones can undergo nucleophilic addition reactions.
Applications[edit]
Pyrones are important in various fields, including:
- Pharmaceuticals: Some pyrone derivatives have biological activity and are used in the development of drugs.
- Flavors and fragrances: Pyrones contribute to the aroma and flavor of certain foods and are used in the fragrance industry.
Examples of Pyrones[edit]
Several naturally occurring and synthetic pyrones are of interest, including:
- Coumarin: A naturally occurring α-pyrone found in many plants.
- Kojic acid: A γ-pyrone derivative used in cosmetics and food products.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
