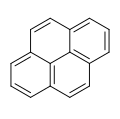
Pyrene
Pyrene is a polycyclic aromatic hydrocarbon (PAH) consisting of four fused benzene rings, resulting in a flat aromatic system. The chemical formula is C16H10. This yellow crystalline solid is the smallest peri-fused PAH (one where the rings are fused through more than one face). Pyrene forms during incomplete combustion of organic compounds.
Properties[edit]
Pyrene is a flat molecule that can intercalate into DNA, while it is not a particularly potent mutagen, it is quite toxic and teratogenic. Pyrene is also used as a probe to determine the polarity of a solvent. It also has a high quantum yield of fluorescence, and because of this it is used in biochemistry experiments as a fluorescent probe, particularly when studying lipids.
Occurrence[edit]
Pyrene and its derivatives are products of combustion and are produced by fossil fuel burning for heat and power, making them common air pollutants. Pyrene is not a major constituent of tar, but it is a component of soot, which is a byproduct of the combustion of diesel and other fuels.
Uses[edit]
Pyrene is used commercially to make dyes and dye precursors, and it is also used in research as a reference standard for the spectroscopic quantification of polycyclic aromatic hydrocarbons.
Health effects[edit]
Exposure to pyrene can cause skin irritation and inflammation. Long-term or repeated exposures can lead to more serious health effects, including cancer.
See also[edit]
Pyrene[edit]
-
Pyrene structure
-
Br4Py self-assembly on gold
-
Br4Py self-assembly on gold (second image)
-
Pyrene numbering
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian