Phytol
Phytol[edit]
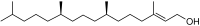
Phytol is an acyclic diterpene alcohol that is an important component in the biosynthesis of chlorophyll and vitamin E. It is a colorless oil with a floral aroma and is used in the fragrance industry as well as in the synthesis of vitamins and other bioactive compounds.
Structure and Properties[edit]
Phytol is a branched-chain unsaturated alcohol with the chemical formula C20H40O. It is characterized by its long hydrophobic chain, which contributes to its role in biological systems. The molecule consists of a 20-carbon chain with a hydroxyl group at one end, making it an alcohol.
Biosynthesis[edit]
Phytol is derived from the degradation of chlorophyll, where it is released from the chlorophyll molecule during the breakdown process. In plants, phytol is esterified to form chlorophyll a and chlorophyll b, which are essential for photosynthesis.
Biological Role[edit]
In addition to its role in chlorophyll, phytol is a precursor for the synthesis of vitamin E (tocopherols and tocotrienols) and vitamin K. These vitamins are crucial for various biological functions, including antioxidant activity and blood coagulation.
Industrial Applications[edit]
Phytol is used in the fragrance industry due to its pleasant aroma. It is also utilized in the synthesis of vitamins and other bioactive compounds. Its derivatives are used in the production of cosmetics, detergents, and pharmaceuticals.
Related Compounds[edit]
Phytol is related to other terpenes and terpenoids, which are a large and diverse class of organic compounds produced by a variety of plants. These compounds are often involved in plant defense mechanisms and have various applications in industry and medicine.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian