Nitrocyclohexane
Nitrocyclohexane[edit]
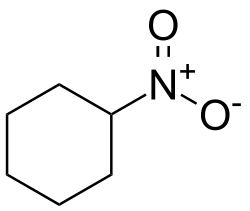
Nitrocyclohexane is an organic compound with the chemical formula C_H__NO_. It is a nitroalkane derivative of cyclohexane, where one hydrogen atom is replaced by a nitro group (-NO_). This compound is of interest in various chemical research and industrial applications due to its unique properties.
Structure and Properties[edit]
Nitrocyclohexane consists of a six-membered carbon ring, typical of cyclohexane, with a nitro group attached to one of the carbon atoms. The presence of the nitro group significantly alters the chemical properties of the molecule compared to its parent hydrocarbon.
The nitro group is electron-withdrawing, which affects the reactivity of the compound. Nitrocyclohexane is a colorless liquid at room temperature and has a characteristic odor. It is relatively stable but can undergo reactions typical of nitro compounds, such as reduction and nitration.
Synthesis[edit]
Nitrocyclohexane can be synthesized through the nitration of cyclohexane. This process involves the reaction of cyclohexane with a nitrating agent, such as nitric acid, under controlled conditions to introduce the nitro group into the cyclohexane ring.
Applications[edit]
Nitrocyclohexane is used in organic synthesis as an intermediate. It can be further transformed into various other chemical compounds, making it valuable in the production of pharmaceuticals, agrochemicals, and other industrial chemicals.
Safety and Handling[edit]
As with many nitro compounds, nitrocyclohexane should be handled with care. It is flammable and can pose health risks if inhaled or ingested. Proper safety measures, including the use of personal protective equipment and adequate ventilation, should be employed when working with this chemical.
Related Compounds[edit]
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian