Methylene (compound)
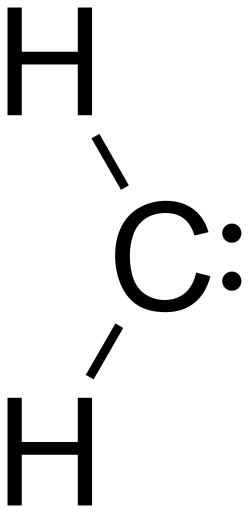
Methylene is a chemical compound with the formula CH2. It is the simplest carbene, a type of reactive intermediate in organic chemistry that contains a carbon atom with only two bonds and two non-bonding electrons. Methylene plays a crucial role in various chemical reactions, including polymerization, cycloaddition, and as an intermediate in the photosynthesis process in plants.
Structure and Properties[edit]
Methylene exists in two major forms: singlet and triplet methylene. The singlet state is a ground state with paired electrons, making it less reactive. In contrast, the triplet state has unpaired electrons, making it highly reactive and a key player in many chemical reactions. The structure of methylene is characterized by its bent shape, with a bond angle of approximately 102° in the singlet state and 136° in the triplet state.
Reactivity and Applications[edit]
Methylene is highly reactive due to its electron configuration. In the triplet state, it can insert into C-H and C-C bonds, making it valuable in organic synthesis. It is also involved in the formation of larger hydrocarbons and is a precursor to many polymers and plastics.
In photosynthesis, methylene is an intermediate in the conversion of carbon dioxide to oxygen and organic compounds, highlighting its importance in biological processes.
Synthesis[edit]
Methylene can be generated in the laboratory through various methods, including the decomposition of certain diazomethane compounds or through the photolysis of ketenes. These methods allow for the controlled study of methylene's reactivity and its application in synthetic chemistry.
Safety and Environmental Impact[edit]
Due to its high reactivity, methylene is not found in significant amounts in the environment and is typically generated in situ for research or industrial purposes. However, its precursors and products should be handled with care due to potential toxicity and environmental impact.
Conclusion[edit]
Methylene, despite its simplicity, plays a significant role in both synthetic and natural chemical processes. Its reactivity and versatility make it a subject of ongoing research in the field of chemistry, with potential applications in the synthesis of complex organic compounds and materials.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
